Advancing medicine and agriculture using proteins that control energy and sugar production
Except for bacteria , most living organisms contain specialized cellular compartments termed mitochondria , that are responsible for breaking down sugars to produce energy for the cell. Plant cells possess a second compartment called chloroplasts , which make sugar (i.e. food) for the plant that can then be converted into energy by mitochondria. These compartments, called organelles, perform important roles in cells and their dysfunction has serious consequences for the organism. In humans, mitochondrial dysfunction can result in genetic disorders such as Parkinson’s disease or inherited deafness. In plants, mitochondrial or chloroplast dysfunction often leads to male sterility and defective embryo development.
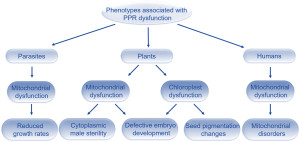
Fig. 1. Summary of defects that can occur in humans, animals and parasites when PPR proteins do not function correctly.
Mitochondria and chloroplast have their own DNA, which encodes genes with the information to produce organelle proteins. These organelle proteins are involved in making energy (in mitochondria) or sugar (in chloroplasts). In recent years, it has become very clear that pentatricopeptide repeat (PPR) proteins are involved in the production of organelle proteins, supported by the fact that defects in PPR proteins leads to organelle dysfunction.
Extensive research has shown that PPR proteins function by either helping or preventing the production of organelle proteins in a highly specific fashion. For example, PPR protein A can control the production of organelle protein A but not organelle protein B. In contrast, PPR protein B can facilitate the synthesis of organelle protein B, but not A. Researchers were perplexed for quite some time about how this specificity occurs, but have recently cracked the code. PPR proteins control organelle protein expression by binding and modifying RNA, messenger molecules that carry the information from the DNA, which is decoded to make organelle proteins. Each PPR protein recognizes a specific RNA sequence that corresponds to the organelle protein of interest, which is why they are highly specific.
This groundbreaking discovery has opened up a whole world of possible applications of these proteins in medicine and agriculture. We are now able to manipulate or design PPR proteins that recognize any RNA sequence we want. These synthetic PPR proteins can be used to replace a dysfunctional PPR protein or even prevent viral infections by having the protein bind and block the synthesis of viral RNA.
Recent work has also shown that the PPR genes have a history of being exchanged between different organisms. This led to the discovery of a whole new family of PPR proteins, PPR-TGM proteins, that are missing in plants and animals. Instead, PPR-TGM genes are found in single-celled organisms like parasites and amoebae, and are derived from bacterial genes. Researchers are investigating whether drugs can be developed to block PPR-TGM function in parasites to induce mitochondrial dysfunction. This is highly desirable as the absence of PPR-TGM proteins in humans means that drugs targeting these proteins effectively treat parasitic infections, but will not have any effects on the human body.
It is evident that PPR proteins are a highly diverse and dynamic group of proteins with important roles in mitochondrial and chloroplast function. Further research into these proteins will improve our ability to utilize the features of PPR proteins to our advantage for curing genetic and infectious diseases, as well as improving crop yields and plant breeding practices.
Sam Manna
Murdoch Childrens Research Institute
Royal Children’s Hospital
Parkville, Victoria, Australia
Publication
An overview of pentatricopeptide repeat proteins and their applications.
Manna S.
Biochimie. 2015 Jun













Leave a Reply
You must be logged in to post a comment.