Bacillus coagulans Unique IS-2 helps ease symptoms of Irritable Bowel Syndrome
Irritable bowel syndrome (IBS) is a common disorder that affects the digestive system. It is a mixture of abdominal pain and altered bowel frequency either constipation, diarrhoea or both (mixed). There is also an alteration in stool consistency (thin, hard, or soft and liquid). Though IBS is not life threatening, it affects the quality of life. The exact cause of IBS is not known, it is multifactorial with change in gut microbiota attributed to be one of the causes. This is where probiotics step in as they help in restoration of normal gut microbiota. The mechanism of action of probiotics include inhibition of colonization by pathogens, supporting the integrity of intestinal epithelial barrier (repairing a leaky gut) and modulation of the immune system. Bacillus coagulans Unique IS-2 is a well characterized, spore forming, safe probiotic strain with efficacy proven in the treatment of diarrhoea, constipation, bacterial vaginosis and IBS in children. In this double blind, placebo controlled trial carried out to study the efficacy of Bacillus coagulans Unique IS-2 in the treatment of IBS in adults, 136 patients ( 18-60 years) diagnosed with constipation related IBS (C) and fulfilling Rome III criteria were divided equally into two groups , the probiotic and placebo groups.
Both the groups were provided placebo capsules for a 2 weeks run-in period after which the patients received either B. coagulans Unique IS-2 (2 billion CFU) or placebo capsules daily for 8 weeks depending upon the groups they were allocated to. Follow up was done for an additional 2 weeks (up to Week 10). The primary parameters assessed were decrease in abdominal pain and increase in spontaneous bowel movements. Other symptoms of IBS like bloating, passage of gas, urgency, feeling of incomplete evacuation, straining etc were assessed through severity of symptoms score which were recorded in the patient’s diary.
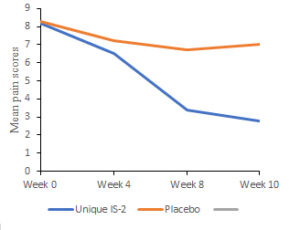
Results: Abdominal pain as assessed by pain intensity scores were significantly reduced ( p < 0.001) in the B.coagulans Unique IS-2 treated group as compared to placebo and baseline (Fig. 1) At the end of treatment , the mean score of abdominal pain was reduced from 8.2 ± 1.37 to 3.4 ± 2.08 in the B. coagulans Unique IS-2 treated group whereas, in placebo , the decrease was marginal i.e. from 8.3 ± 1.25 to 6.7 ± 1.92. Patients showing ≥ 50% pain reduction from their baseline visit were considered as responders in the study. By the end of treatment, 84.91% of the patients in the B. coagulans Unique IS-2 treated group demonstrated ≥ 50% pain reduction compared to only 12.73% (p < 0.001) patients in the placebo treated group. Complete bowel spontaneous movements (CBSM) were also significantly increased (p < 0.001 ) in 79.25% of B. coagulans Unique IS-2 treated group whereas in the placebo group there was an increment of only 40.00 % by the end of treatment (8 weeks). The same trend was observed at the follow up period which was after two weeks i.e.10th week (Fig. 2) Significant improvement (p < 0.0001) in other symptoms of IBS like abdominal discomfort, bloating, urgency, incomplete evacuation, straining, passage of gas and bowel habit satisfaction were also noted in the B. coagulans Unique IS-2 treated group from the 5th week onwards.
No adverse effects were observed in either of the groups.
Conclusion: Bacillus coagulans Unique IS-2 significantly improved symptoms of IBS in adults and can be taken by patients to help alleviate symptoms.
Ratna Sudha Madempudi, Jayesh J. Ahire, Jayanthi Neelamraju
Centre for Research & Development, Unique Biotech Ltd., Plot No. 2, Phase-II,
Alexandria Knowledge Park, Hyderabad, Telangana, 500078, India
Publication
Randomized clinical trial: the effect of probiotic Bacillus coagulans Unique IS2 vs. placebo on the symptoms management of irritable bowel syndrome in adults
Ratna Sudha Madempudi , Jayesh J. Ahire, Jayanthi Neelamraju, Anirudh Tripathi, Satyavrat Nanal
Sci Rep. 2019 Aug 21














Leave a Reply
You must be logged in to post a comment.