Cs adsorption by Mn‒Fe-based Prussian blue analogs formed in agarose gel
The Fukushima nuclear accident triggered a massive release of radioactive cesium (Cs) isotopes into the environment and generated a large amount of contaminated water. Because Cs isotopes pose serious threats to the environment (particularly the aquatic environment), selective, efficient, and inexpensive sorbents for Cs+ ions in solutions are highly desired. Currently, the most promising Cs sorbents are Prussian blue (PB) and their analogs (PBAs). For instance, PB has been approved as a drug used in clinics for the treatment of radioactive Cs exposure.

Fig. 1. (A) A snapshot of the Mn‒Fe PBA precipitates spontaneously formed in agarose gel and (B) a SEM image of the precipitates, obtained at magnifications of 5000. (C) Depicting the Cs adsorption at the Mn‒Fe PBAs in agarose gel.
PB and PBAs exhibit a wide variety of compositions, crystal structures, and Cs adsorption abilities, and these properties strongly depend on the reaction conditions and preparation methods. In a recent study published in the Journal of Analytical Atomic Spectrometry (2019, 34, 979–985), we reported that the precipitation bands of Mn‒Fe-based PBAs (Mn‒Fe PBAs) in agarose gels trap Cs+ ions more effectively than those of PB. In this study, such strong adsorption of Cs+ ions by the Mn‒Fe PBAs was investigated by extended X-ray absorption fine structure (EXAFS) spectroscopy coupled with scanning electron microscopy (SEM). For these measurements, gel samples were prepared by contacting a gel containing 0.05 M [Fe(CN)6]3− and 2.3 mass% agarose with a 0.50 M MnSO4 solution, into which a 0.10 M CsCl solution was introduced. The Mn‒Fe PBA precipitates spontaneously form relatively broad, periodic banding structures in agarose gel (Fig. 1A). The SEM revealed that the Mn‒Fe PBA precipitates consist of aggregates of cubic crystallites that bear considerable amounts of defects (Fig. 1B).
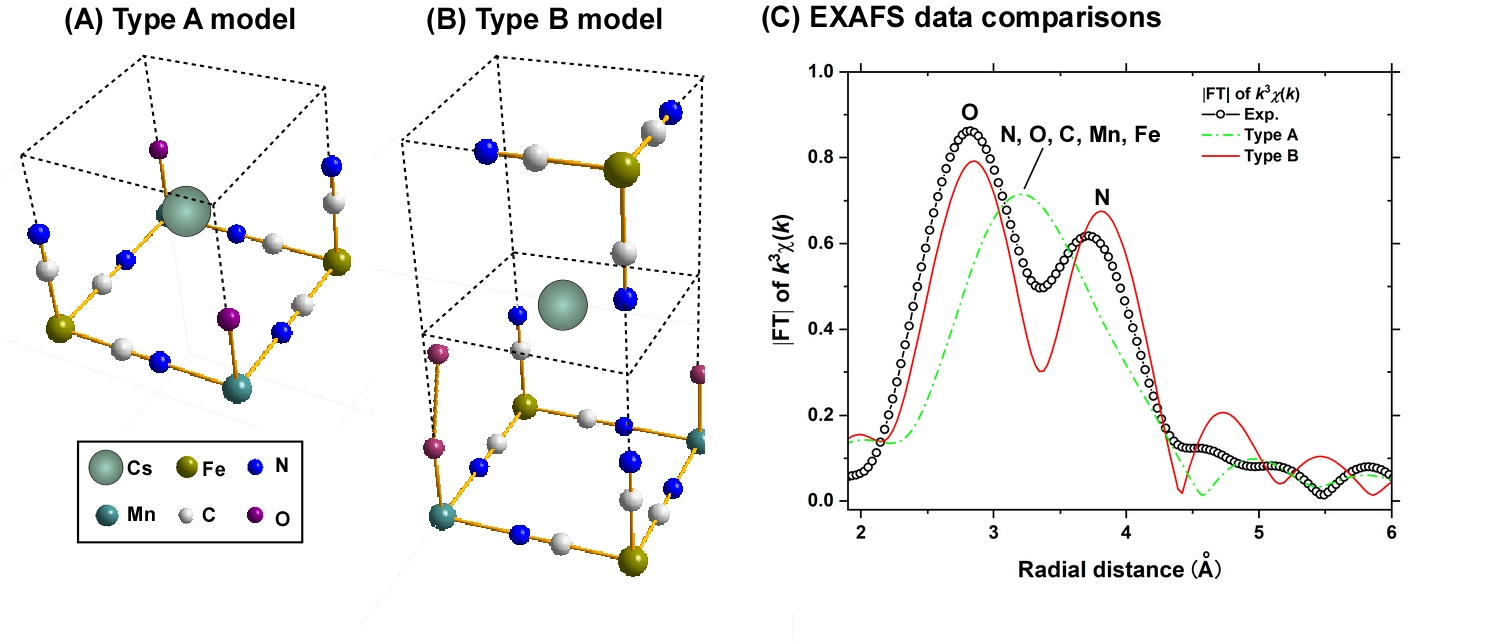
Fig. 2. Depicting (A) the type-A and (B) type-B cluster models used to analyze (C) the Cs L3-edge EXAFS data.
Figure 2A and B display cluster models used to analyze the Cs L3-edge EXAFS profile. In the type A cluster, a Cs+ ion is situated at the body-center of the cube (cubic center) constructed by the Mn‒N‒C‒Fe moieties, which are partially replaced by O atoms (conceivably H2O molecules). In the type B cluster, a Cs+ ion is localized at the center of a cube face, being surrounded by the vacant sites. The differences between these models are evident in the Fourier transform (FT) of the EXAFS oscillation (k3χ(k); Fig. 2C). The FT spectrum based on the type A shows only a broad single peak, making it impossible to assign the characteristic two peaks of the experimental FT spectrum (Exp). In contrast, the FT spectrum based on the type B reproduces the two experimental peaks fairly well, attributing the high and low peaks to the first-nearest O and the second-nearest N atoms, respectively, strongly suggesting that the Mn‒Fe PBA crystallites mainly adsorb Cs+ ions at large defect sites close to the cube faces. In accordance with this finding, the Mn K-edge XANES results show that many cubic centers in the Mn‒Fe PBAs are mainly filled with H2O molecules or K+ ions prior to Cs adsorption, and these molecules/ions are mostly displaced during Cs adsorption. The suggested Cs adsorption mechanism (Fig. 1C) provides valuable first insight toward fully understanding Cs adsorption by Mn‒Fe PBAs.
Hisashi Hayashi
Department of Chemical and Biological Sciences, Faculty of Science, Japan Women’s University, Japan
Publication
An XAFS study of Cs adsorption by the precipitation bands of Mn-Fe-based Prussian blue analogues spontaneously formed in agarose gel
Hisashi Hayashi, Saya Aoki, Mao Takaishi, Yui Sato, Hitoshi Abe
Phys Chem Chem Phys. 2019 Oct 28













Leave a Reply
You must be logged in to post a comment.