Does acute kidney injury affect other organs?
Acute Kidney Injury (AKI) is a common and serious condition with no specific treatment. AKI has been estimated to cost the NHS in the UK alone between £451-626 million per year. Recovery from a single episode of AKI is known to then increase risk of being diagnosed with chronic kidney disease later in life. However, many patients that recover from AKI also appear to be a greater risk of developing certain diseases of other organs distant or ‘remote’ from the kidney such as the lung or liver. Why acute damage to the kidney may affect other organs is not known but it is thought that other organs may become infiltrated with inflammatory cells which affect their function in the longer term. Nearly all experimental studies to date on the remote effects of AKI have been conducted in laboratory rodents (e.g. rat and mouse), which may not be clinically valid. Whilst clinical studies are preferred for investigating human disease – humans are, after all, the best experimental animal model for humans – clinical studies of AKI are often confounded by multiple comorbidities and tissue sampling of remote organs like the lung is difficult, risky and usually unethical. Epidemiological studies can suggest associations between AKI and effects on remote organs, but will not help understand the cause of the problem. Pigs are excellent biomedical models for human disease, especially renal disease. Here, we used a large animal (porcine) model of AKI to investigate any potential remote effects.
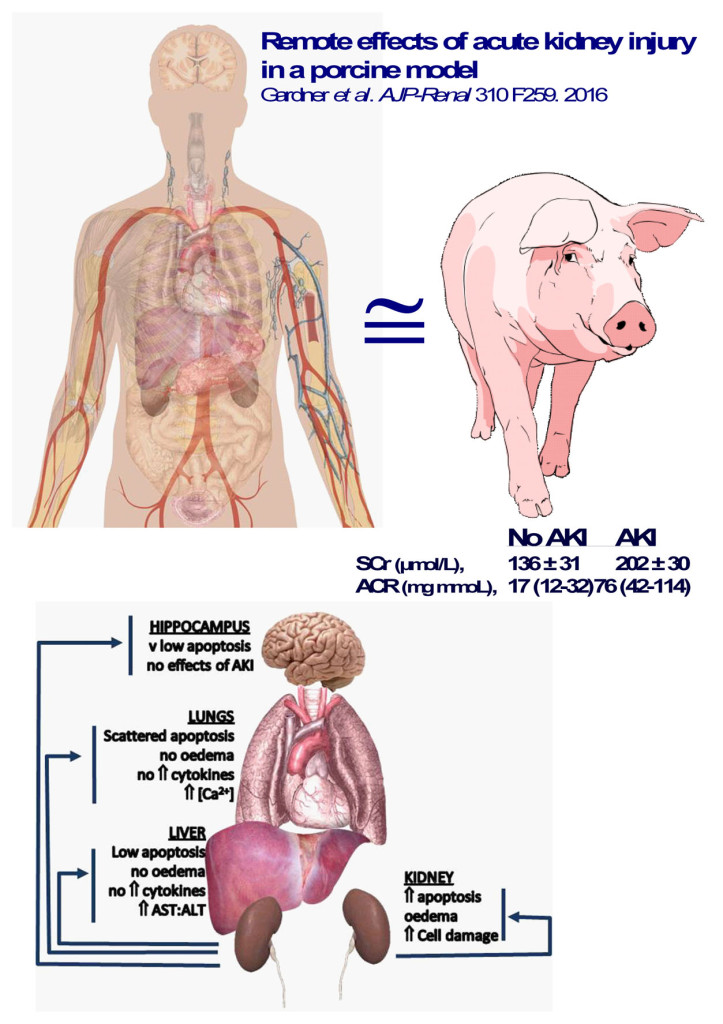 We first characterized a time-course of immune cell activation after whole blood sampling (0-24h) using hematology and flow-cytometry and then assessed possible immune cell infiltration in kidney and other organs (liver, lung and brain-hippocampus) using a variety of laboratory techniques. Two veterinary pathologists, blinded to the experimental design, independently scored the level of injury in the remote organs. At 48h after moderate ischemic AKI we observed no overt histopathology, fluid accumulation or indices of cell death as a result of inflammatory cell infiltration in either the liver, lung or hippocampus. In the liver, we did pick up some indices of short-term damage and in lung, some indices of altered tissue chemistry. However, the size of these effects were biologically small and likely to be experimental artefacts. Longer-term follow-up of these animals indicated that these effects had not worsened and that all experimental animals had recovered from the acute kidney injury to a satisfactory extent.
We first characterized a time-course of immune cell activation after whole blood sampling (0-24h) using hematology and flow-cytometry and then assessed possible immune cell infiltration in kidney and other organs (liver, lung and brain-hippocampus) using a variety of laboratory techniques. Two veterinary pathologists, blinded to the experimental design, independently scored the level of injury in the remote organs. At 48h after moderate ischemic AKI we observed no overt histopathology, fluid accumulation or indices of cell death as a result of inflammatory cell infiltration in either the liver, lung or hippocampus. In the liver, we did pick up some indices of short-term damage and in lung, some indices of altered tissue chemistry. However, the size of these effects were biologically small and likely to be experimental artefacts. Longer-term follow-up of these animals indicated that these effects had not worsened and that all experimental animals had recovered from the acute kidney injury to a satisfactory extent.
Warm ischemia-reperfusion AKI, sufficiently moderate to damage kidney cells and elevate plasma creatinine by 100-200%, had subtle biochemical effects on some remote organs (e.g. lung and liver). The effects were, however, mild and did not get worse with time. We suggest that the moderate and sometimes marked remote organ effects seen in laboratory rodent models may not reflect the clinical picture adequately. Our study reiterates that 1) choice of animal model is very important for most pre-clinical studies, especially in the renal field and 2) for potential translation into clinical practice in general. It is likely that remote effects may occur after isolated, acute kidney injury but only in conjunction with significant other co-morbidities such as disease status or advanced age.
Dr. David S Gardner, BSc, PhD, DSc
Associate Professor in Developmental Physiology
University of Nottingham, Faculty of Medicine and Health Sciences
School of Veterinary Medicine and Science, Sutton Bonington Campus
Loughborough, UK
Publication
Remote effects of acute kidney injury in a porcine model.
Gardner DS, De Brot S, Dunford LJ, Grau-Roma L, Welham SJ, Fallman R, O’Sullivan SE, Oh W, Devonald MA.
Am J Physiol Renal Physiol. 2016 Feb 15













Leave a Reply
You must be logged in to post a comment.