Exploring early mechanotransduction responses
Mechanical stress plays a key role in cellular behaviour and fate, affecting the correct development and functionality of tissues and organs. Due to the complexity of the in vivo cellular environment, most mechanotransduction research relies on the development of in vitro techniques able to dissect cell responses to specific mechanical stimuli. In this perspective, in vitro cell-stretching assays represent the main approach for further understanding the dynamics of mechanotransduction.
Stretch-induced responses, which generally occur at the cell surface in the proximity of sites of mechano-sensation, emerge at very short time scales such as seconds or minutes. These can include opening of stretch-activated cation channels, ions fluxes, induction of enzymatic activity of integrin-associated signalling molecules, induction of heterotrimeric G-proteins and production of reactive oxygen species.
The development of innovative research tools is therefore of the utmost importance to help unravel the complicated interconnection between mechano-sensing elements and biochemical pathways within the cell. How can we reproduce this phenomena in vitro and capture their very early effects on cell behaviour?
First of all, the investigation of such mechanisms occurring during or immediately after an imposed static or dynamic mechanical stimulus requires a real time imaging of the cells during the exposure to mechanical stretch. Secondly, the possibility to perturb the soluble environment without disturbing the real time cell imaging would be needed.
Several devices to stretch cells are currently available and, among them, miniaturized systems based on microfluidics are the ones that guarantee an accurate control of the cellular microenvironment, thanks to the low volumes (nL to uL) and timing of fluid manipulation. However, stretching devices completely based on the microfluidic technology allowing an easy coupling with microscopes for real time analysis have been not completely exploited yet.
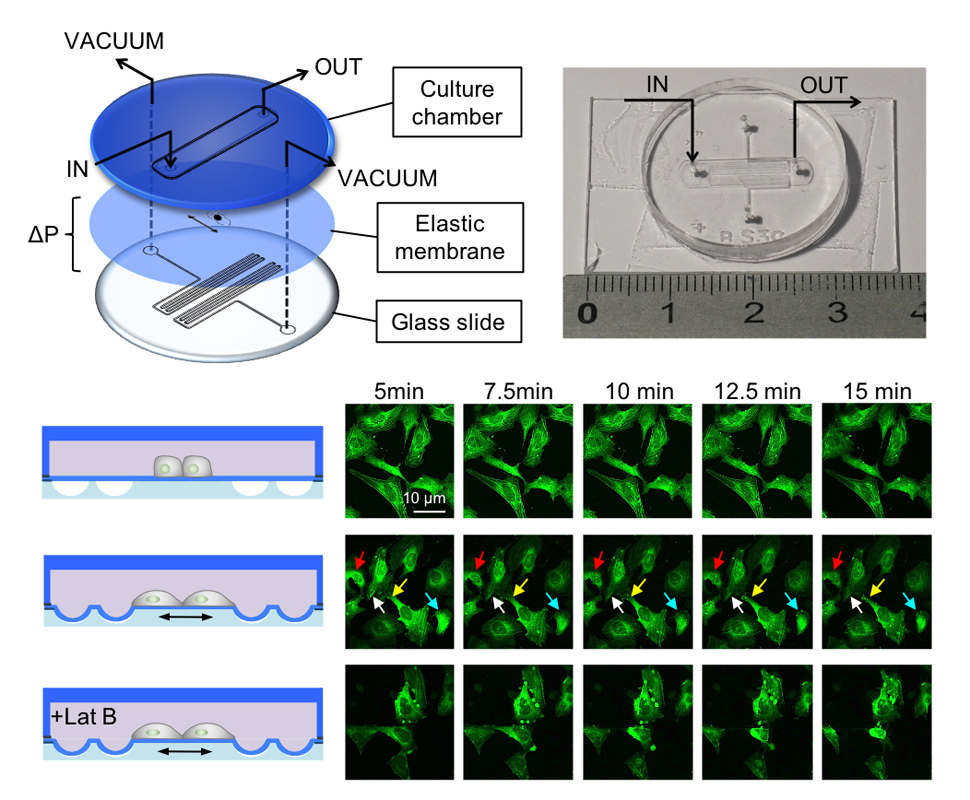
We developed a novel multi-layered microfluidic platform that allows accurate mechanical stimulations of cells while maintaining the focal plane during real-time high-resolution confocal imaging. The platform is coupled with a microfluidic cell culture chamber with the purpose of accurately controlling the soluble microenvironment in terms of biochemical composition, while allowing mechanical stimuli and fast analysis of mechano-transduction-associated cell responses (Fig. 1). Such a system is ideal to study rapid events in response to mechanical stretch, in particular those related to conformational changes of cell components, such as membrane or cytoskeletal tension.
The design of the device was optimized using numerical simulation to identify the correlation between working parameters, such as membrane thickness, channels shape and applied pressure difference, and cell deformation. The microfluidic device is fully biocompatible and is able to stretch cultured cells up to 20% total area deformation, showing a linear correlation between deformation and applied pressure difference. The cell deformation can be easily tuned by varying the device design and the applied pressure difference. This allows exploring both physiological and pathological strain conditions and instantaneous responses can be easily analysed in more than one cells at the same time, thus increasing the readout significance.
Taking advantage of the developed device we imaged actin filaments remodelling, by means of a fluorescent reporter cell line, in real-time during the first minutes of stretching, observing a increasing remodelling of the actin filaments network. The analysis was performed in the presence or absence of Latrunculin B, a potent inhibitor of actin polymerization that disrupt filament organization.
Overall, the developed device allows the unique combination of accurate cell mechanical stimulation, real-time microscope analysis at high magnification while maintaining the focal plane and possibility to accurately control the soluble microenvironment thanks to automatic and precise medium delivery inside the microfluidic cell culture chamber. This will further support the increasing research in the field of mechanotransduction.
Federica Michielin, Nicola Elvassore
University of Padova, Padova, Italy
Publication
A Novel Microfluidic Platform for Biomechano-Stimulations on a Chip.
Prevedello L, Michielin F, Balcon M, Savio E, Pavan P, Elvassore N
Ann Biomed Eng. 2019 Jan













Leave a Reply
You must be logged in to post a comment.