How does chromium cause lung cancer? Resistance to cellular stress
Lung cancer is the deadliest form of cancer in both men and women. One cause of lung cancer is occupational exposure to heavy metals. Extremely useful in several industries, heavy metals have become widespread environmental pollutants over the last decades. Yet, the mechanisms of heavy metal-induced carcinogenesis remain poorly understood.
We focused on chromium (Cr), an important component of stainless steel and limb prosthetics. Chromium exists in two main oxidation states: Cr3+ or Cr(III) and Cr6+ or Cr(VI). The latter enters cells via the phosphate transporter channel (chromium and phosphate ions have very similar tetrahedral structures and sizes). Once inside cells, Cr(VI) gets successively reduced to Cr(III), in the process cross-linking DNA and proteins and releasing reactive oxygen species (ROS). These nefarious byproducts are believed to be responsible for most of Cr(VI) toxicity. Their involvement in cell transformation into cancer cells, however, is unclear.
To simulate occupational exposure to Cr(VI) in the laboratory, we treated human lung epithelial cells (BEAS-2B) with very low doses (1 μM) of Cr(VI). This is an important aspect, as too high concentrations of Cr(VI) are unlikely to mimic environmental exposure and are overtly toxic, leading to cell death rather than cell transformation.
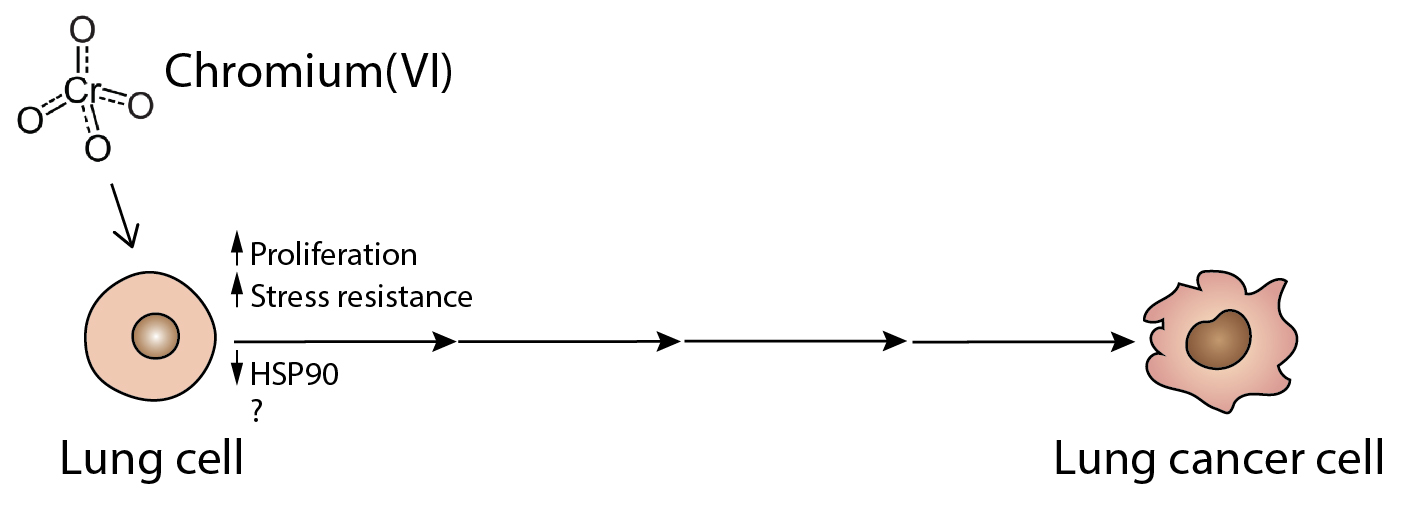
We found that these low doses increased the rate of cell division. Interestingly, low dose Cr(VI) also protected cells from thermal shock-induced reduction in the rate of cell division. Carcinogenesis can be seen as an evolutionary process, where incipient cancer cells get selected based on their capacity to withstand stress caused by factors such as nutrient and oxygen deprivation, DNA damage, and protein misfolding. One way in which cells cope with stress is by invoking the heat shock response. Key components of this pathway are heat shock proteins (HSPs). These proteins act as chaperones to assist with protein folding; excessive amounts of misfolded proteins inside a cell will lead to endoplasmic reticulum (ER) stress and, if left unchecked, cell death. HSP activity has thus been found to be crucial in rapidly proliferating cells, in particular cancer cells, which not only divide ferociously but also produce a higher amount of mutant proteins that require extra chaperoning to fold properly. HSP90 has a vast array of client proteins, which combined are involved in virtually every hallmark of cancer. Indeed, HSP90 is the heat shock protein most commonly found to be upregulated in cancer.
Yet, to our surprise, low dose Cr(VI) caused a decrease in the levels of HSP90 protein in treated cells. Moreover, HSP70, another heat shock protein, was not modulated by Cr(VI) at this dose. HSP70 has been suggested to take over some of HSP90 functions when the latter is therapeutically inhibited. Many questions thus remain unanswered. Are other HSPs being modulated by Cr(VI) at these very early stages? Or is it a completely novel mechanism of resistance to stress? And how do the levels of these proteins change as we move along the path from healthy to fully malignant lung cancer cell? We will continue to push forward on these and other experiments to better understand and ultimately treat lung cancer.
Leonardo M.R. Ferreira
Department of Surgery, Transplantation Research Laboratory
Diabetes Center, Sean N. Parker Autoimmune Research Laboratory
University of California, San Francisco, San Francisco, CA, USA
Publication
Hexavalent chromium, a lung carcinogen, confers resistance to thermal stress and interferes with heat shock protein expression in human bronchial epithelial cells.
Abreu PL, Cunha-Oliveira T, Ferreira LMR, Urbano AM
Biometals. 2018 Aug













Leave a Reply
You must be logged in to post a comment.