Innovative tool, promise for treating Alzheimer’s disease and other neurodegenerative brain diseases
If at the dawn of the twentieth century, Alzheimer’s disease was seen as a rare disease, but today the incidence of this disease has taken epidemic proportions and is treated as one of the diseases of civilization. The development of digital technology frees people from the need to strain your brain to solve routine tasks. Creative trends, activating brain functions are replaced by more and more consumer. Just as our body grows old from a lack of physical activity, and our brains – from the lack of a kind of mental gymnastics. All this is compounded by physical inactivity and chronic stress.
The essence of Alzheimer’s disease is gradually dying nerve cells of the brain, whereby the patients lose their memory and mental abilities increasingly weak. Developing senile dementia, speech disorder, apathy, impaired self-service patient. In public offering of economic damage Alzheimer’s takes “honorable” third place (after heart disease and cancer pathology). Analysts consulting firm GlobalData predict a rise in the global market of drugs for the treatment of Alzheimer’s disease to 13.3 billion US dollars by 2023. In 2013 it was equal to the amount of 4.9 billion US dollars.
One of the leading causes of Alzheimer’s disease – the adverse effects of excess primary transmitter of nerve impulses in the brain, glutamate. Glutamate target cell surface are receptors that perceive glutamate signals. Overexcitation glutamate receptors leads to the fact that an excessive amount of calcium enters in the neuron cells and this leads to neuronal death. Rises task – how to prevent this process?
In 2002, Merz developed Memantine – the first effective drug used worldwide for the treatment of moderately severe cases of dementia caused by Alzheimer’s disease. Its unique mechanism of action protects neurons from high concentrations of glutamate. Memantine has certain advantages over other known ‘antiglutamate” drugs, because blocks is not an absolute, but only the excessive activation of glutamate receptor and, therefore, is generally well tolerated. Memantine is the most frequently purchased agent for treatment Alzheimer’s disease in the world.
Knowledge of the mechanism of action of memantine allows us to raise the question of a wider range of application – not only for the treatment of dementia, but also for the protection of neurons in other neurodegenerative diseases (Parkinson’s disease, epilepsy, chorea). Universal neuroprotective action of memantine may be achieved with doses 2-3 times higher than the doses for treatment of patients with Alzheimer’s disease.
However, a small therapeutic range of action of memantine does not allow to realize these opportunities. Even a slight excess standard therapeutic dose of memantine leads to side effects such as sleep disturbance, motor coordination, qualmishness, dizziness, aggressiveness, motor stimulation and in some cases, there is a development of psychosis.
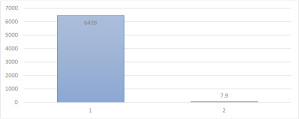
We have synthesized and investigated new glutamate blocker, a structural analogue of Memantine, under the code IEM-1913, which in animal experiments considerably exceeds the activity of memantine on models of diseases such as Parkinson’s, epilepsy, depression, pain, and most importantly, IEM-1913 much safer compared to memantine. In contrast, memantine, only at the maximum effective and safe for use doses (10-20 mg/kg), IEM-1913 causes significant pharmacological effects over a wide dose range 0.03-1 mg/kg without any side effects. As a result, IEM-1913 has a very high therapeutic index equal to 6430 (memantine – 7.9). Thus, IEM-1913 has a therapeutic index in 814 times greater than memantine (Fig. 1.), and therefore, in contrast to memantine, the compound is substantially non-toxic and safe for use.
Publication
Comparison of Pharmacological Potency and Safety of Glutamate Blocker IEM-1913 and Memantine.
Gmiro VE, Serdyuk SE, Veselkina OS
Bull Exp Biol Med. 2015 Nov













Leave a Reply
You must be logged in to post a comment.