Macrophage-myofibroblast transition: a novel target for stopping kidney disease
Kidney is one of the major organs in our body for detoxification. Its failure is an important cause of patient mortality that serves as a primary disease and a lethal complication in a range of diseases such as diabetes, cancer, bacterial and virus infections including COVID-19; resulting in more than 6 million patients receive renal replacement therapy worldwide. It is well known that kidney fibrosis is a key pathological feature and common pathway leading to the loss of normal kidney structure and functions, resulting in chronic kidney failure. However, this urgent area of clinical need is still unmet due to the lack of effective treatment to stop the process of kidney scarring. Encouragingly, scientists have started to recognize that hyperactivation of our immune system is critical for promoting this process.
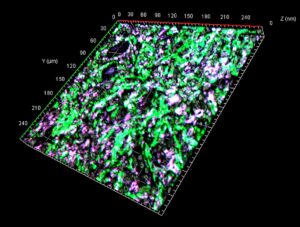
Fig. 1. The presence of MMT in a diseased kidney of patient with chronic allograft dysfunction (Tang et al., PNAS 2020;117:34:20741-20752).
Macrophage is a well-documented immune cell type for maintaining the health of our kidneys. They are a type of white blood cell that serves as a soldier in our body, responsible for detecting, engulfing and destroying pathogens and unhealthy cells. Paradoxically, new studies found that their preponderance also accelerates organ failure. A better understanding of the underlying mechanisms would separate out the unwanted actions of macrophages form a diseased kidney. Therefore, we attempted to dissect the dynamics of macrophages in the diseased kidney with a latest analytic tool singe-cell RNA-sequencing. Surprisingly, our study discovered that long-term hyperactivation of macrophages will direct their transformation into a scar-producing cell type called myofibroblasts in the diseased kidney, which promotes tissue scarring via a novel phenomenon ‘Macrophage–Myofibroblast Transition (MMT).
Importantly, we confirmed that MMT cells are abundant in the patient biopsies, which may represent a critical step for turning a healthy kidney to loss function. Moreover, we further identified a brain-specific gene POU4F1 is essential for establishing MMT in the diseased kidney by reconstructing the regulatory gene network of macrophages at single-cell resolution with an unbiased bioinformatic approach. We found that abnormal expression of POU4F1, a transcription factor for sensory neurons development, in the kidney-infiltrating macrophages is essential for initiating MMT. Inspired by these findings, related preclinical works have been started by treating two mouse kidney disease models with gene therapy. Encouragingly, our results showed that macrophage-specific silencing of POU4F1 effectively prevented tissue scarring by blocking the generation of myofibroblasts from MMT and therefore stopping kidney failure.

Fig. 2. POU4F1 is a potential therapeutic target for blocking MMT-driven kidney diseases (Image modified from Tang et al., Nature Reviews Nephrology 2019;15(3):144-158).
Immunotherapy shows promise in cancer treatment and its implications in other diseases has begun to be explored. Our study has identified a new immune regulator for promoting organ failure and developed a novel immunotherapeutic strategy to protect patients against kidney disease. This study was supported by the CU Medicine Faculty Innovation Award, Hong Kong Research Grants Council, Health and Medical Research Fund, Innovation and Technology Fund, Guangdong-Hong Kong-Macao-Joint Labs Programme from the Guangdong Science and Technology Department and Lui Che Woo Institute of Innovative Medicine (CARE programme). The finding has been published in the prestigious international scientific journal Proceedings of the National Academy of Sciences.
Patrick Ming-Kuen Tang 1, Hui-Yao Lan 2
1Department of Anatomical and Cellular Pathology, State Key Laboratory of Translational Oncology, Prince of Wales Hospital, The Chinese University of Hong Kong.
2Department of Medicine & Therapeutics, Li Ka Shing Institute of Health Sciences, Lui Che Woo Institute of Innovative Medicine, The Chinese University of Hong Kong
Publication
Neural transcription factor Pou4f1 promotes renal fibrosis via macrophage-myofibroblast transition
Patrick Ming-Kuen Tang, Ying-Ying Zhang, Jun Xiao, Philip Chiu-Tsun Tang, Jeff Yat-Fai Chung, Jinhong Li, Vivian Weiwen Xue, Xiao-Ru Huang, Charing Ching-Ning Chong, Chi-Fai Ng, Tin-Lap Lee, Ka-Fai To, David J Nikolic-Paterson, Hui-Yao Lan
Proc Natl Acad Sci U S A. 2020 Aug 25













Leave a Reply
You must be logged in to post a comment.