Nanomaterials as promising alternative in the infection treatment
Nanotechnology offers groundbreaking solutions for the management of infections in the current era when the vast majority of antibiotics has become inefficient. Overcoming antimicrobial resistance and biofilm formation constitute a major challenge. In this review, we focus on the recent scientific advances to design inorganic-based nanomaterials from two different approaches: preventing infection or combating it once stablished (Fig. 1).

Fig. 1 .Two common infection-fighting strategies based on the design of nanostructured materials. The strategy to inhibit bacterial adhesion via surface modification is shown on the left. The use of nanosystems to destroy the formed biofilm using nanoparticles with intrinsic antimicrobial properties and nanoparticles acting as nanocarriers of different of antimicrobial agents is shown on the right.
The term “nano”, according to the FDA and IUPAC, refers to any product with properties or phenomena attributable to its dimensions, when such dimensions are in the nanoscale range of 1–100 nm. Nanomaterials have unique properties since they exhibit a large surface area to volume ratio and high versatility, which could enhance their influence on a given pathogenic microorganism. Thus, the advantage of these nanomaterial formulations over conventional systems is that they can increase treatment efficacy and decrease side effects through their precise targeting mode of action.
Regarding prevention of infection, the inhibition of bacterial adhesion is a key aspect in bone implant-devices. Tailoring the biomaterial surface has been tackled through two main strategies: i) chemical modification to create zwitterionic surfaces, which bear equal numbers of positive and negative charges (overall electrical neutrality), therefore showing high resistance to nonspecific protein adsorption, bacterial adhesion and biofilm formation; ii) textural modifications to tailor the nanotopography and nanostructure surface, mimicking those found in Nature such as leaves lotus and insect wings. The creation of a dense and highly packed nanotopography provides a superhydrophobic surface that inhibits bacterial adhesion and subsequent biofilm formation.
Combating infection with nanomaterials have been described from two flanks, i.e., nanoparticles with inherent antibacterial activity and those that are vehicles to deliver antimicrobial agents.
Antibacterial nanoparticles such as metal (Au, Ag, etc.) and metal oxide nanoparticles (ZnO, Fe3O4, CuO, MgO and TiO2) act against bacteria through mechanisms which are related to oxidative stress, metal ion release, and non-oxidative mechanisms, and generally trigger the formation of reactive oxygen species (ROS), enzymatic inhibition, protein deactivation, DNA damage, or changes in gene expression as well as bacteria wall disruption. The advantage of using these antimicrobial nanoparticles is that their multiple mechanisms of action differ from the standard mechanisms of antibiotics, making them extremely useful against bacterial infection avoiding the dreaded antimicrobial resistance.
Among nanomaterials as vehicles for antimicrobial agents, mesoporous silica nanoparticles (MSNs) constitute a promising advanced inorganic nanoplatform for drug delivery. The main strengths of MSNs are high loading capacity, biocompatibility, ease of production, and high degree of tunability regarding size, morphology, and pore diameter. Furthermore, MSNs can be easily synthesized on a large scale, showing a great variety of morphologies and surface functionalities using different strategies. Figure 2 shows a schematic representation of the versatility and functionality of MSNs for biomedical applications.
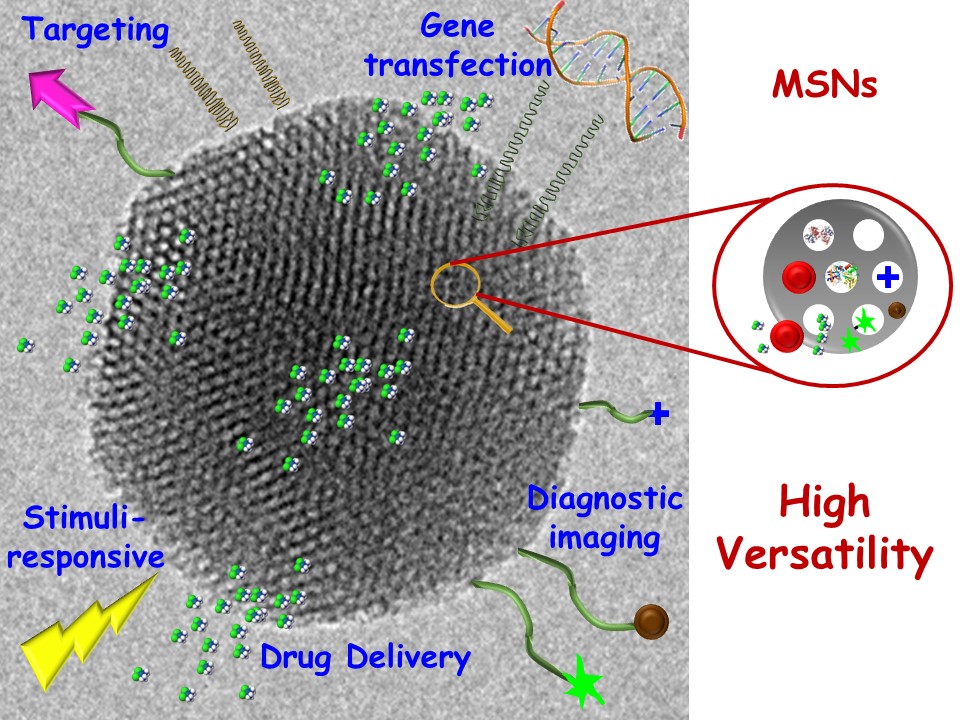
Fig. 2. Schematic representation of the versatility and functionality of mesoporous silica nanoparticles (MSNs). TEM image corresponds to an MSN of ca. 150 nm in diameter showing the mesoporous arrangement in the 2D hexagonal structure (p6mm plain group). The cartoons represent the drug loading capability, active targeting, and stimuli-responsive possibilities of MSNs.
In this review we highlight the main achievements in the design and development of MSNs taking advantage of their targeting capability to combat the infection. Our pioneering research work has focused on developing innovative MSNs-based systems thorough surface functionalization with targeting moieties towards the bacteria and/or biofilm, so a local high concentration of the drug and considerably increase the activity of the loaded antimicrobial agent can be achieved at the infection site. Thus, decorating the external surface of MSNs with macromolecules such as polycationic dendrimers or lectins, and loading the mesopores with antibiotic molecules allowed the design of a troy horse capable to penetrate the bacterial wall or bacterial biofilm matrix. Therefore, the internalization in the targeted site significantly increased the antimicrobial effectiveness of the loaded antibiotic.
This is certainly the starting point towards a considerable improvement in conventional treatments, where the tendency is to combine all the elements in order to effectively abolish the dreaded infections.
María Vallet-Regí, Montserrat Colilla, Blanca González, Isabel Izquierdo-Barba
Departamento de Química en Ciencias Farmacéuticas, Unidad de Química Inorgánica y Bioinorgánica, Universidad Complutense de Madrid,
Instituto de Investigación Sanitaria Hospital 12 de Octubre i+12, Plaza Ramón y Cajal s/n, Madrid 28040, Spain
CIBER de Bioingeniería, Biomateriales y Nanomedicina, CIBER-BBN, Madrid 28040, Spain
Publication
Nanomaterials as Promising Alternative in the Infection Treatment
María Vallet-Regí, Blanca González, Isabel Izquierdo-Barba
Int J Mol Sci. 2019 Aug 4













Leave a Reply
You must be logged in to post a comment.