Osteoarthritis alleviation by rewiring cellular energy metabolism
Osteoarthritis (OA) is a chronic disorder associated with degeneration of the joint, including articular cartilage, subchondral bone, synovial tissue, tendons, periarticular muscles that involves low-grade systemic inflammation. Recent studies have demonstrated the involvement of chondrocyte differentiation (hypertrophy) as one of the mechanisms in cartilage degradation in OA. This finding suggests common regulatory mechanisms that govern chondrocyte differentiation and articular cartilage destruction in OA. As cell proliferation and differentiation are regulated by the cellular metabolic status related to energy production, here we suggest that knowledge of the mechanisms required for maintenance of energy homeostasis could provide a means to reveal the primary causes of OA development.
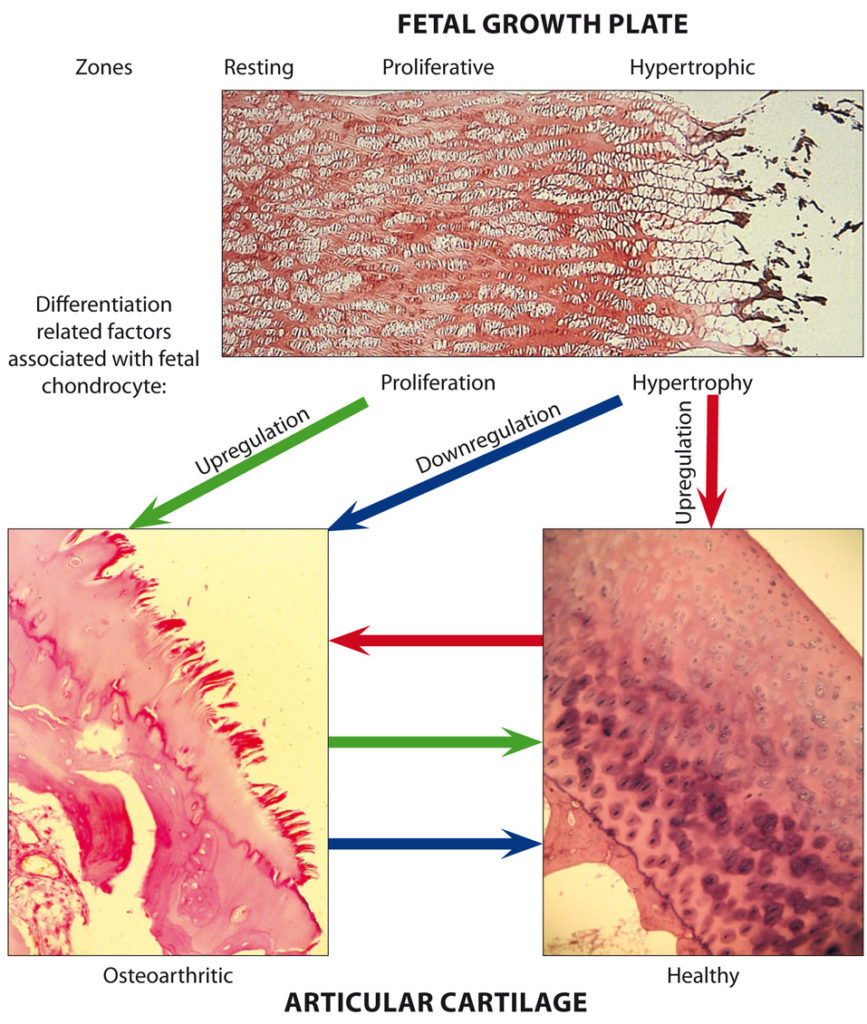
Fig. 1. Mechanisms of articular cartilage destruction in osteoarthritis.
Fetal growth plate is divided into zones: resting, proliferative, and hypertrophic. OA-related changes in healthy articular cartilage occur on upregulation of fetal chondrocyte hypertrophy-associated differentiation factors (red arrows). In contrast, upregulation of fetal chondrocyte proliferation-associated factors (green arrows) and/or downregulation of hypertrophy-associated differentiation factors (blue arrows) in OA articular cartilage result in suppression of OA-related traits producing healthier phenotype.
Eukaryotic cells can generate energy in the form of ATP either using the anaerobic process of glycolysis producing two ATP molecules or by aerobic oxidative phosphorylation (OXPHOS) generating up to 30 ATP molecules. The OXPHOS system integrates the production of cellular energy with major catabolic pathways, including the tricarboxylic acid (TCA) cycle and fatty acid or amino acid oxidation. It contains five multimeric mitochondrial proteins: Complexes I-V. The chain of redox reactions produces an electrochemical gradient, which is used by Complex V to drive ATP synthesis.
Intracellular regulation of energy production is principally important for differentiation during normal growth. Multiple signalling pathways affect both proliferation and differentiation and control cellular energy metabolism. Anabolic signalling pathways such as mammalian target of rapamycin (mTOR) mostly stimulate glycolysis. AMP-activated protein kinase (AMPK) promotes catabolic activity and fatty acid oxidation, inhibits glycolysis, and upregulates the TCA cycle.
Endochondral ossification (Fig. 1), the mechanism responsible for the development of long bones, is dependent on an extremely stringent coordination between the processes of chondrocyte maturation in the growth plate, vascular expansion in the surrounding tissues, osteoblast differentiation, and osteogenesis. Growth plate cartilage is a highly metabolic but poorly vascularized tissue adapted to generating energy primarily by glycolysis. However, the increase in the mitochondrial protein fraction and the decrease in mitochondrial volume observed in resting cartilage compared to the hypertrophic zone indicates that residing chondrocytes change their energy status during endochondral ossification.
In OA (Fig. 1), inflammatory mediators induce mitochondrial dysfunction and increased production of reactive oxygen species. Oxidative stress might also damage the chondrocyte mitochondrial respiratory chain, as evidenced by the decreased activity of Complexes I, II, and III, causing decreased mitochondrial ATP generation. Increased glycolysis activity suggests that OA chondrocytes from anti-inflammatory reliance on oxidative phosphorylation and the TCA cycle switch to proinflammatory reliance on glycolysis for energy.
In addition, the metabolic syndrome components such as abdominal obesity, hypertension, hyperglycaemia, significantly increase the risk of disturbances to normal functioning of energy and nutrient sensors in articular cartilage. At the molecular level, metabolic dysregulation in OA was associated with reduced activities of AMPK as well as increased mTOR expression in articular chondrocytes.
Furthermore, reduced AMPK gene expression in OA articular cartilage was accompanied by its increased expression in the peripheral blood cells from the same OA patients. This suggests that whole-body energy availability and that in articular cartilage can differ in OA patients and cause an energy appeal reaction, a process that involves the redirection of energy-rich fuels from energy sources, such as articular cartilage in OA for further energy relocation to other specific body sites in the course of its destruction. Indeed, the upregulation of collagen type II expression in OA cartilage with concomitant upregulation of TCA-related gene expressions points to the potential for availability of energy-generating substrates required for matrix repair by chondrocytes in OA patients.
Elena Tchetina
Nasonova Research Institute of Rheumatology, Moscow, Russia
Publication
Regulation of energy metabolism in the growth plate and osteoarthritic chondrocytes.
Tchetina EV, Markova GA
Rheumatol Int. 2018 Nov
Related Articles:
 | Unlocking new treatments for bone diseases: using… Despite appearances, bones are a constantly changing organ in the body. Cells that produce new bone (named osteoblasts) and cells that break down bone (named osteoclasts) work in harmony to… |
 | PEPITEM supports new bone and blood vessel growth in… While bones may appear solid and unchanging, they are a living tissue that is constantly being broken down and rebuilt in a process known as bone remodelling. Specialised cells (called… |
 | Systemic therapy in juvenile localized scleroderma Juvenile localized scleroderma, also known as morphea, is an rare condition, it occurs around 1 in 5000 children, where the skin and the tissues underneath the skin like fat tissue,… |
 | Looking inside the heart: how multiple chronic… The aim of this study was to understand how having several ongoing health problems—what we refer to as multimorbidity—impacts the heart in people with cardiovascular disease, especially those undergoing heart… |
 | Could mometasone furoate be a viable glucocorticoid… Glucocorticoids are medications widely used to treat inflammation and allergies, and in contexts of immunosuppression such as after transplants. They are well-established and safe. The problem is that, when used… |
 | Naturally occurring peptide may tackle the “root… Obesity typically occurs when a person eats more calories than they need for their daily activity. It results in a build-up of fatty tissue around all organs. In lean people,… |







Leave a Reply
You must be logged in to post a comment.