Summary of: Screening for viral hepatitis prior to rituximab chemotherapy
There are an estimated 800,000-1.4 million people with Hepatitis B Virus (HBV) in the US. Patients with a low immune systems, such as cancer patients, are at an increased risk of reactivation of HBV if they have been infected prior to chemotherapy. When the immune system is highly suppressed by specific chemotherapy the virus is allowed to replicate, but when the chemotherapy is stopped and the immune system returns the immune system attacks the virus in the liver. Rituximab is one of the drugs associated with HBV reactivation.
 In 2008, the CDC recommended that patients receiving cytotoxic or immunosuppressive therapy undergo testing for HBV infection. In September 2013, the FDA added a warning for rituximab stating there was a risk of reactivation of HBV infection. They recommended HBV screening prior to rituximab initiation and discontinuation of rituximab if HBV reactivation occured while on therapy. There have been only a few clinical studies done to determine the clinical application of the guidelines recommending HBV screening prior to initiating chemotherapy in institutional settings.
In 2008, the CDC recommended that patients receiving cytotoxic or immunosuppressive therapy undergo testing for HBV infection. In September 2013, the FDA added a warning for rituximab stating there was a risk of reactivation of HBV infection. They recommended HBV screening prior to rituximab initiation and discontinuation of rituximab if HBV reactivation occured while on therapy. There have been only a few clinical studies done to determine the clinical application of the guidelines recommending HBV screening prior to initiating chemotherapy in institutional settings.
This study included patients ≥18 years of age whom received rituximab between January 2006 and December 2012 at one institution. The main goal of this study was to compare HBV screening rates for veteran patients who received rituximab based chemotherapy prior to and after the release of the 2008 CDC guideline to see if there was a significant increase in screening. We also looked to determine if risk factors for hepatitis infection predicted hepatitis screening. The patients were divided into patients that received rituximab between 2006-2009 and 2010-2012. These dates were chosen to allow time for knowledge of the 2008 CDC guideline spread through the medical community
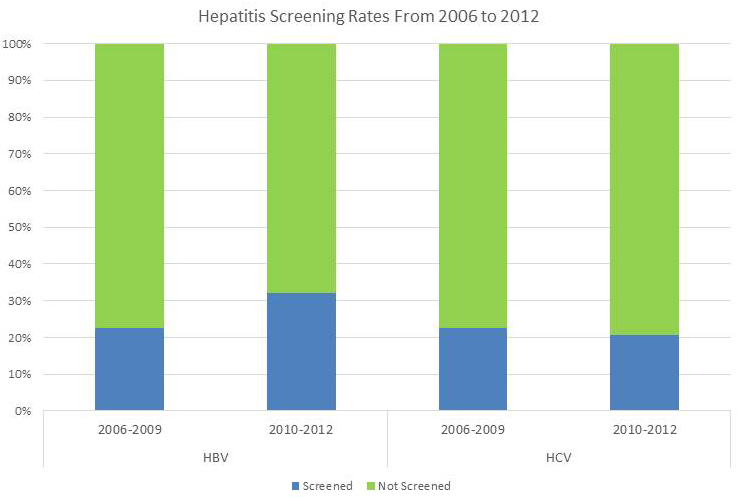
From 2006-2012 a total of 102 patients were treated with rituximab. Our study population was 97% male. Of the common HBV risk factors, medical records documented that 12 patients had history of substance/alcohol abuse, 1 patient was homeless, 9 patients had a diagnosis of Hepatitis C Virus (HCV), and 3 patients had a diagnosis of HIV. From 2006-2009, 11 of 49 (22%) patients were screened for HBV infection compared with 17 of 53 (32%) patients from 2010-2012. For HCV, from 2006-2009, 11 of 49 (22%) patients were screened compared with 11 of 53 (21%) patients from 2010-2012.
We found that the baseline rate of HBV screening was low (22%) prior to dissemination of the CDC guidelines and did not consistently increase after the CDC guidelines were disseminated. In 2009, there was an initial increase in only that year. We believe this was likely due to heighted awareness of the need to screen patients. We also found that, HCV screening of the same rituximab-treated Veterans, had similarly low screening results. There were also low rates of HBV and HCV screening among Veterans patients with established risk factors (substance/alcohol abuse, HIV, homelessness) for HBV and HCV infection. With an increased risk for both HBV and HCV infections veterans are at a very high risk for hepatitis reactivation if they are not screened and given immunosuppressive therapy.
There are several possible quality improvement initiatives that could be considered to improve HBV screening prior to immunosuppressive chemotherapy. One tool that is readily available for VA medical centers would be to create an EMR template that would require a negative HBV test before rituximab could be initiated.
HBV reactivation has the potential to be fatal, but it is also preventable if screened for as recommended. Effective quality improvement tools need to be implemented and evaluated to ensure that rituximab-treated patients are appropriately screened for HBV.
Publication
Screening for viral hepatitis prior to rituximab chemotherapy.
Leonard AN, Love BL, Norris LB, Siddiqui SK, Wallam MN, Bennett CL
Ann Hematol. 2015 Sep 18.
Related Articles:
 | Shutting down unwanted visitors: Please be quiet Our cells respond to invading DNAs formed by viral infection in a variety of ways to suppress their ability to produce RNAs and proteins. One mechanism is to wrap the… |
 | Lipid nanoparticles loaded with CpG oligonucleotides… Since the emergence of immune checkpoint inhibitors (ICIs), cancer immunotherapy has dramatically developed. However, its drawback is the low overall response rate to its therapeutic effect. The efficacy of immunotherapy… |
 | Is multiple sclerosis triggered by immunological… Multiple sclerosis (MS) is an autoimmune disease where immune cells (T cells) and antibodies progressively damage the myelin sheath surrounding nerve cells leading to their loss of function. We have… |
 | Naturally occurring peptide may tackle the “root… Obesity typically occurs when a person eats more calories than they need for their daily activity. It results in a build-up of fatty tissue around all organs. In lean people,… |
 | Improving assessment of arthritis models to better… Rheumatoid Arthritis (RA) is a common inflammatory disease that is characterized by swelling and tenderness of multiple joints. The resulting pain and joint stiffness cause disability for patients and treatment… |
 | Rabbits with mammary carcinomas as a model for… Within a breeding colony of rabbits, the American pathologist Harry Greene (1904-1969) observed that mammary carcinomas were restricted to certain families. This is suggestive of a familiar predisposition as it… |







Leave a Reply
You must be logged in to post a comment.