Targeted GRP78 pathway for improved vision in diabetic retinopathy
Diabetic retinopathy, is a complication of diabetes which causes vision loss and eventual blindness in adults. People with diabetes have more sugar in their blood than normal, which causes their bodies not to function correctly. Often proteins coated with sugars through a process called O-GlcNAcylation play a vital role in the healthy cells. Because the body needs the correct balance of proteins to be healthy, any increase or decrease in O-GlcNAcylation could potentially cause damage.
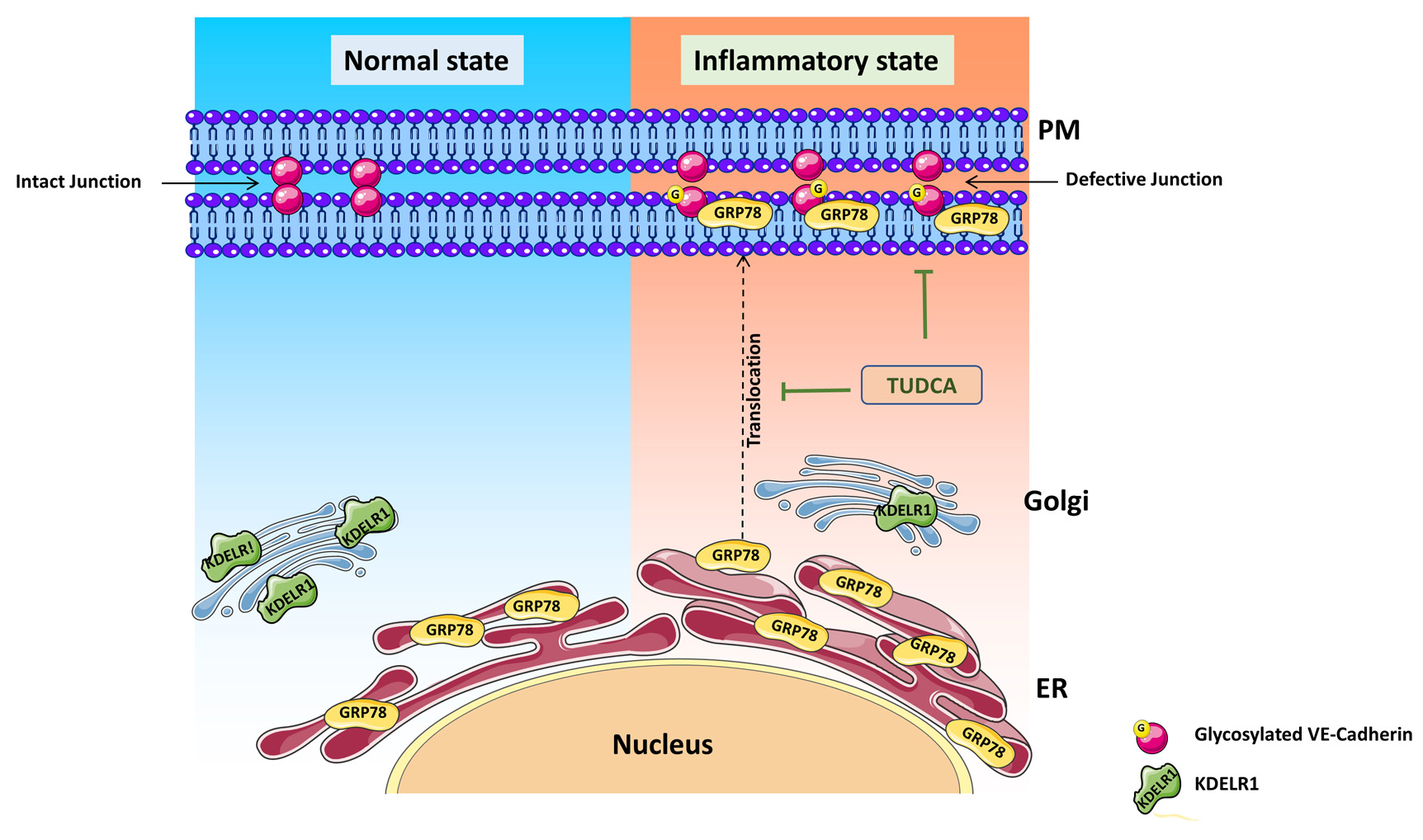
Fig. 1. Schematic model of GRP78 translocation to the plasma membrane (PM) under ER stress leading to retinal endothelial permeability. In pathological state such as chronic inflammation and hyperglycemia will lead to up-regulation of ER stress in retinal endothelial cells. In the absence of KDELR-1, GRP78 translocates to the cell surface accentuating ER stress and O-GlcNAcylation of proteins. O-glycosylation of endothelial junction protein (VE-Cadherin) likely to form defective junctions leading to endothelial hyperpermeability. Inhibition of ER stress by tauroursodeoxycholic acid (TUDCA) protects against O-GlcNAcylation changes thus protects barrier integrity. Created with content from Servier Medical Art (https://smart.servier.com) under Creative Commons Attribution 3.0 Unported license (https://creativecommons.org/licenses/by/3.0/).
The cells lining the blood vessels (endothelium) in the eye have specialized systems of protein synthesis that help the eye’s blood vessels stay healthy. In the endothelium, endoplasmic reticulum (ER) an intracellular organelle is responsible for proper protein folding and maturation including O-GlcNAcylation. Previous studies have shown that inflammation mediated damage to retinal vasculature leads to dysfunctional ER leading to a phenomenon called ER stress, however, the exact mechanisms of how diabetes, inflammation and ER stress are linked to blood vessel damage is not known. Using endothelial cells from eyes incubated in a dish with high sugar and pro-inflammatory cytokine, TNF-α, we found that a ER specific protein in our body called glucose-regulated protein (GRP78) was activated, resulting in ER stress. GRP78 has an ER retention signal called as KDEL peptide, but when the KDEL is absent, the GRP78 could move out of the ER. We confirmed that in endothelial cells exposed to high sugar and inflammation leads to the transfer of GRP78 out of the ER to the plasma membrane of the cells. At the plasma membrane, we showed that GRP78 interacts with the VE-Cadherin, a junction protein in the endothelium that is required for cell-to-cell adhesion in the blood vessels augmenting the addition of sugar to VE-Cadherin (GlcNAcylated VE-cadherin). Interestingly O-GlcNAcylation of junction proteins further resulted in increased transmigration of leukocytes across a monolayer of endothelium and increased permeability under these stress conditions creating a perpetual motion of inflammation and ER stress.
By studying the interaction of these proteins that lead to leaky blood vessels, one can hope to find a drug that can prevent the blood vessels from developing ER stress and O-GlcNAcylation in vascular endothelium. To this end, we showed that both O-GlcNAcylation inhibitors and ER stress inhibitor-TUDCA, a bile acid derivative can prevent inflammation and high sugar induced increased transmigration of leukocytes and increased permeability in the endothelium. Our findings suggest an important role for ER stress and O-GlcNAcylation in altering the endothelial function and reveal a potential therapeutic target to treat the blood vessel damage in the eye.
Scientists have recently found that people with cancer have similar increased levels of GRP78, though it is unclear if this increase is a cause or effect of the disease. Since GRP78 respond to ER stress and likely affect cell function in diabetic endothelium as well as in cancers, targeting GRP78 pathway for diabetic retinopathy may hold important value as also being explored for cancer therapies. Future research needs to establish cause and effect relationship of GRP78 translocation to the plasma membrane to dysfunctional endothelium in diabetic models for developing such targeted therapies.
Raji Lenin, Rajashekhar Gangaraju
Dept of Pharmacology and Nutritional Sciences, University of Kentucky, Lexington, KY, USA
Dept of Ophthalmology, Dept of Anatomy & Neurobiology, University of Tennessee Health Science Center, Memphis, TN, USA
Publication
GRP78 translocation to the cell surface and O-GlcNAcylation of VE-Cadherin contribute to ER stress-mediated endothelial permeability
Lenin R, Nagy PG, Jha KA, Gangaraju R
Sci Rep. 2019 Jul 25













Leave a Reply
You must be logged in to post a comment.