Therapy of osteoporosis by the transdermal formulation based on raloxifene nanoparticles in ovariectomized rat
Osteoporosis results from insufficient post-menopausal estrogen levels and collapse of the balance between osteoclasts and osteoblasts, and the osteoporosis is a common systemic skeletal condition that is related to low bone mineral density and pathological fractures. The main pharmacologic agents used for treatment are anti-resorptives and raloxifene hydrochloride (raloxifene) that prevent the action and development of osteoclasts. The raloxifene is a benzothiophene derivative, classified pharmacologically as a selective estrogen receptor modulator, and is effective for the prevention and treatment of osteoporosis. It is mostly supplied as 60 mg tablets as the daily dose for patients, and is approved for the prevention and treatment of post-menopausal osteoporosis. However, the raloxifene belongs to Biopharmaceutical Classification System (BCS) class II drugs, and the absolute bioavailability (BA) of raloxifene in oral administration is approximately 1.7% due to its poor water solubility and extensive first-pass metabolism. Therefore, it is hypothesized that raloxifene could be a more effective drug for the prevention and treatment of osteoporosis if its BA could be enhanced.
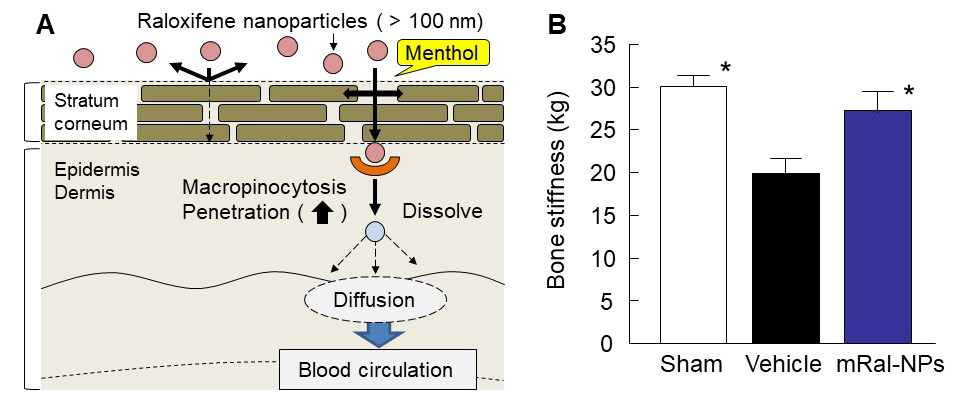
Fig. 1. Transdermal penetration pathway and therapeutic effect in the combination of raloxifene nanoparticles and menthol. (A) Mechanism for transdermal penetration via macropinocytosis. (B) Changes in bone stiffness in OVX rats treated with 0.3% mRal-NPs. Means ± S.E. n=5-8. *P<0.05 vs. Vehicle for each category.
A transdermal delivery system offers a way to avoid first-pass metabolism and drug-drug interactions, and any techniques to enhance drug delivery through transdermal routes have been studied, such as nanoparticles. However, it is difficult for particles larger than 100 nm to penetrate through the stratum corneum, and the production of nanoparticles under 100 nm take much cost. Taken together, we propose a transdermal formulation containing nanoparticles (100 nm – 450 nm) and permeation enhancers (menthol, a terpene alcohol), and demonstrated whether the transdermal formulation is useful are useful for the treatment of osteoporosis.
In general, it is known that passage through the stratum corneum is the rate-limiting step in percutaneous absorption, and presents the greatest resistance to penetration. The infiltration of raloxifene nanoparticles over 100 nm from the transdermal formulations is also impeded by the stratum corneum. On the other hand, 2% menthol preferentially distributes into the intercellular spaces of the stratum corneum thus altering the barrier properties of the stratum corneum and causing a reversible disruption of the lipid domains, and the raloxifene nanoparticles with menthol (mRal-NPs) penetrate the barrier of the stratum corneum, dissolve in the skin tissue, and are released from the skin as soluble raloxifene (Fig. 1A).
It is important to elucidate the mechanism of the transdermal absorption of raloxifene nanoparticles, and it was known that the endocytosis is the major route by which nanomedicines are transported across the membrane. In this study using raloxifene nanoparticles (100 nm – 450 nm), the micropinocytosis, one kind of endocytosis, accounts for the skin penetration of raloxifene nanoparticles, since the raloxifene nanoparticles reach the underlying epidermis via the stratum corneum, and are taken up into cells by micropinocytosis (Fig. 1A).
The therapeutic effect of mRal-NPs on osteoporosis using ovariectomized rats (OVX rat) as a model was also demonstrated. The Ca contents and stiffness in the bones of OVX rats decreased following ovariectomy, and the application of 0.3% mRal-NPs attenuated the decreases in Ca content and stiffness in the bones of OVX rats (Fig. 1B). These results show that mRal-NPs provides useful therapy for osteoporosis.
In conclusion, we found that the menthol attenuates the barrier function of the stratum corneum, allowing raloxifene particles that are approximately 100 nm – 450 nm in size to transit into the underlying epidermis. After that, the raloxifene dissolve and diffuse into the epidermis, dermis, and ultimately be absorbed into the blood circulation (BA 8.5%). The enhanced levels of plasma raloxifene provide efficient and effective therapy for osteoporosis. These findings provide significant information that can be used to design further studies aimed at developing transdermal delivery systems using nanoparticles.
Noriaki Nagai and Naohito Kawasaki
Faculty of Pharmacy, Kindai University, 3-4-1 Kowakae, Higashi-Osaka, Osaka, Japan
Publication
Design of a transdermal formulation containing raloxifene nanoparticles for osteoporosis treatment.
Nagai N, Ogata F, Otake H, Nakazawa Y, Kawasaki N
Int J Nanomedicine. 2018 Sep 6













Leave a Reply
You must be logged in to post a comment.