Tumor regression in a high-mortality mouse pancreatic cancer (xenograft) model with aminosteroid RM-581
Pancreatic cancer is classified as a high-mortality cancer with a five-year relative survival rate of only 9%. Although most cancer type survival rates have improved over the past decades, pancreatic cancer morbidity has been stable, which illustrates the clinical need for new therapeutic tools for the treatment of this fatal disease. Endoplasmic reticulum (ER) is a predominant organelle in pancreatic cancer cells exposed to a high level of stress from a hostile tumor microenvironment. In fact, ER stress induces cellular dysfunction that can lead to apoptosis, which is a form of programmed cell death.
Structure-activity relationship studies on the anticancer activity of a new family of aminosteroid derivatives built around an androstane backbone has demonstrated strong antiproliferative properties on several cancer cells, including human pancreatic cancer cells, as well as cancer tumor models (xenografts) in mice. Despite a positive proof-of-concept in a pancreatic mouse cancer model, the metabolic stability of RM-133 was not optimal for translation to a clinical setting. In a new round of optimization, we improved the pharmacological properties of RM-133 by replacing its androstane (a steroid with 19 carbones) backbone by an estrane one (a steroid with 18 carbones) like the mestranol core. Fortunately, this modification exemplified by the estrane derivative RM-581 (Fig. 1A) advantageously increased metabolic selectivity for cancerous cells over normal cells, without losing anticancer activity.
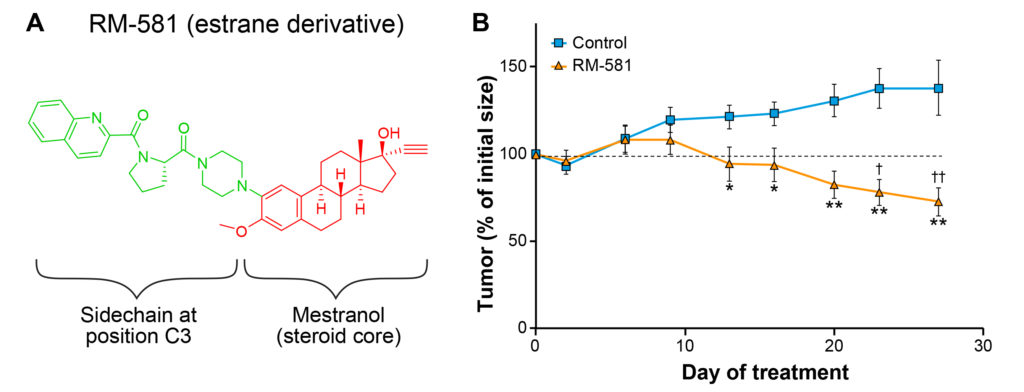
Fig. 1. A) Chemical representation of aminosteroid derivative RM-581 showing its 2 crucial structural elements. B) RM-581 causes a full tumor regression in pancreatic cancer model (xenografts with human PANC-1 cells) when given orally 6 days per week in nude mice. Black square: vehicle alone (propylene glycol and dimethylsulfoxide in proportions 92:8); Green triangle: RM-581 (60 mg/kg dissolved in vehicle).
The aminosteroid RM-581 was found to be significantly more active compared to a series of chemotherapeutic agents used in clinic in 3 cancer cell lines known to be resistant to the chemotherapeutic drug gemcitabine. It was also classified as an ER stress aggravator, inducing similar ER-stress markers in cancer cells as RM-133. Interestingly, a 60 mg/kg dose of RM-581 administered orally resulted in a concentration of 1,860 ± 301 ng/mL, 1 hour after gavage. The blood exposure was also considerable, comforting the idea that RM-581 is bioavailable orally, contrary to RM-133. For comparison, the oral administration of the same dose of these two aminosteroids resulted in a much higher blood concentration for RM-581 (8.5 times), measured 3 hours after dosing. Although the administration schedule or formulation of RM-581 could be further optimized to reach full beneficial effects, these pharmacokinetic results encouraged us to address its ability to block tumor progression in a known model of pancreatic cancer (PANC-1 cells xenograft).
When administered per gavage (orally) 6 days per week in nude mice that developed human pancreatic (PANC-1) tumors (Fig. 1B), RM-581 stopped tumor progression from day 13 and provoked a significant regression in tumor size from day 23, up to an unmeasurable level, due to the small size of the tumors. Interestingly, any sign of toxicity (mouse weight and behavior) was observed during the 27 days of the protocol. We also completed our toxicology evaluation of RM-581’s adverse effects using tissue slice visualization. Histological examination of mouse liver, kidney and intestines, 3 of the organs most affected by drug toxicity, did not show any sign of toxicity after the treatment of mice with RM-581 for 27 days. Together, our in vitro and in vivo toxicological results demonstrate that RM-581 is a selective anticancer molecule for cancer cells over normal cells.
Altogether, these relevant results obtained in cancer cells and successfully translated to nude mice by testing RM-581 at different doses (10-60 mg/kg) and modes of administration using human PANC-1 cell xenografts as cancer model, qualified RM-581 as a valuable preclinical candidate to be further evaluated in pancreatic cancer clinical phase trials. The emergence of the aminosteroid RM-581 as a new therapeutic drug will open the way to treat refractory pancreatic cancers, as well as other types of cancers, through an innovative approach.
Donald Poirier
Department of Molecular Medicine, Faculty of Medicine, Université Laval, Québec, QC, Canada
Laboratory of Medicinal Chemistry, Endocrinology and Nephrology Unit, CHU de Québec – Research Center, Québec, QC, Canada
Publication
Induction of endoplasmic reticulum stress by aminosteroid derivative RM-581 leads to tumor regression in PANC-1 xenograft model.
Perreault M, Maltais R, Roy J, Picard S, Popa I, Bertrand N, Poirier, D
Invest New Drugs. 2019 Jun













Leave a Reply
You must be logged in to post a comment.