What are the chemical properties of water in the world’s deepest blue hole?
The Sansha Yongle Blue Hole (SYBH), located on the Yongle Atoll of the Xiasha Islands in the northern South China Sea, has been discovered to be the world’s deepest blue hole. Its depth is 301.19 m, 100 m deeper than the previous record holder, namely, Dean’s Blue Hole located off the Bahamas. The cross section of the SYBH resembles the shape of a ballet shoe held vertically, with its diameter varying from ~130 m at the surface to ~32 m at the bottom. Although tidally influenced, the water column within the SYBH is protected from open ocean circulation and exhibits diverse physicochemical–microbial gradients. Two thermoclines, in which temperatures decrease more rapidly with depth than in adjacent layers, can be observed, and these divide the water column into the following five distinct layers: upper mixed layer (top 13 m), seasonal thermocline (13–20 m), weak mixing layer (20–70 m), permanent thermocline (70–150 m), and homogeneous deep water (depths of more than 150 m). According to 14C data, modern carbon exists only above a depth of 70 m, which indicates that water is vertically mixed above the permanent thermocline.
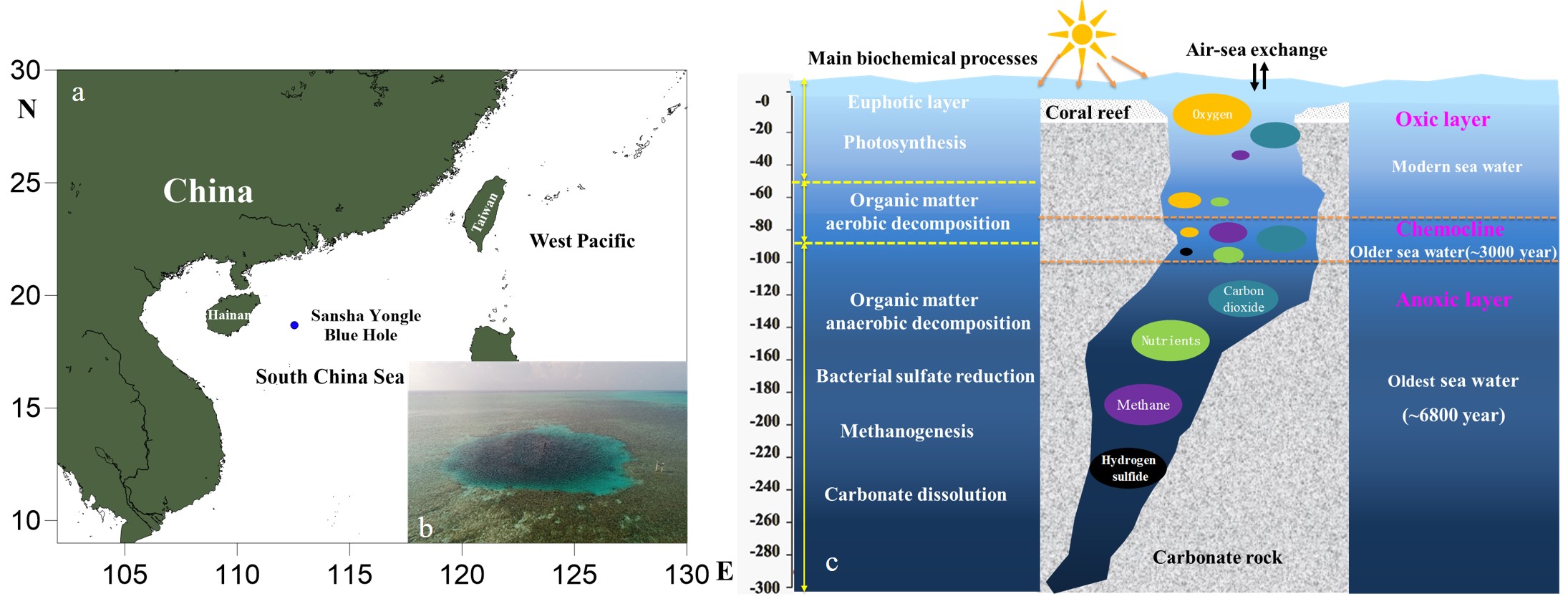
Fig. 1. (a) Location and (b) bird’s-eye view of the Sansha Yongle Blue Hole; (c) variations of biogenic elements and related biochemical processes in different redox systems.
Chemical analyses have shown that there is no significant difference in content of major elements such as Na+, K+, Ca2+, Mg2+, and Cl– between the seawater inside and outside the blue hole. However, different redox systems exist inside the blue hole because of the complex interactions among physical, chemical, and biological processes. On the basis of the redox state, the water column can be divided into the following three layers: oxic water (top 70 m), chemocline (70–100 m), which acts as a redox boundary, and anoxic deep layer (depths of more than 100 m). In the oxic layer, dissolved oxygen is relatively abundant in the top mixed layer because of free air–sea exchanges; here, phytoplankton have access to sunlight and grow well, taking up nutrients and transforming inorganic carbon (i.e., carbon dioxide) into organic carbon through photosynthesis, which results in undetectable levels of nutrients (i.e., nitrate, nitrite, ammonium, phosphate, and silicate). Below the seasonal thermocline, lower solar radiation limits photosynthesis, and decomposition of organic matter is the main chemical–biological process that takes place. This process consumes oxygen and releases nutrients and carbon dioxide, which leads to decreases in dissolved oxygen and pH.
Within the chemocline, organic matter decays via various reactions (e.g., aerobic mineralization, denitrification, and anammox) and there are sharp decreases in oxidizing chemical species (e.g., dissolved oxygen and nitrate) and increases in reducing species (e.g., ammonium and sulfide); microbial mediated denitrification drives sharp decreases in nitrate, but peak values of nitrite and nitrous oxide (a greenhouse gas) and pronounced increases in ammonium concentrations occur at a depth of 90 m. Nitrogen is removed from seawater, whereby ~60% of the nitrogen is lost via denitrification and anammox. Additionally, sharp decreases in pH occur because of the carbon dioxide released during the decomposition of organic matter.
Within the deep anoxic layer, the lowest concentrations and oldest ages (e.g., 6810 years before present) of organic carbon can be observed. Here, there is no dissolved oxygen, but there is abundant hydrogen sulfide, which is highly toxic and smells like rotten eggs. Huge quantities of carbon dioxide accumulate in the water, partly as a result of the decomposition of organic carbon and the dissolution of carbonates. As in other anoxic environments (e.g., the Black Sea and Baltic Sea), there are also high concentrations of methane (so-called marsh gas) and nutrients, among which ammonium is the only form of dissolved inorganic nitrogen.
Linping Xie, Baodong Wang, Tiegang Li
First Institute of Oceanography, Ministry of Natural Resources, Qingdao, China
Publication
Hydrochemical properties and chemocline of the Sansha Yongle Blue Hole in the South China Sea.
Linping Xie, Baodong Wang, Xinming Pu, Ming Xin, Peiqing He, Chengxuan Li, Qinsheng Wei, Xuelei Zhang, Tiegang Li
Sci Total Environ. 2019 Feb 1













Leave a Reply
You must be logged in to post a comment.