Brand new weaponry in our arsenal: nanopatterns to combat biomaterial-associated infections
Humankind has witnessed unprecedented breakthroughs in (bio)medical sciences during the 20th and 21st centuries. A significant increase in life expectancy has cultivated the seeds of eagerness to live longer and healthier in the collective mind of humans. If we succeeded to eradicate smallpox then what would prevent us from annihilating cancers? If we can get a pacemaker, why not a whole personalized 3D-bio-printed heart in the time of need? To what extent can we expand the lifespan?
Well-appreciated questions and enthusiasm but let’s focus on reality. Biological systems are too complex to be easily understood, mimicked, or manipulated. Perhaps in 50 years, we will have totally different answers to the abovementioned questions but at the moment even simpler-looking problems require the hard work of researchers for more comprehensive or newer answers.
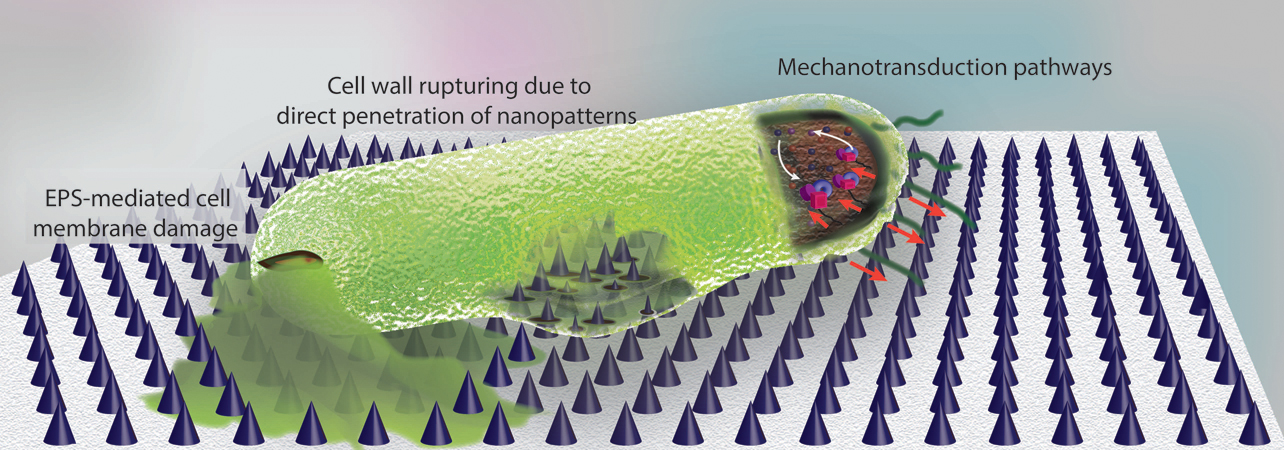
Fig. 1. The main bactericidal mechanisms of nanopatterns. While the commonly believed theory is that bacterial cell wall is ruptured by penetration of high aspect ratio nanopatterns, there are a few studies suggesting that extracellular polymeric substance (EPS) plays a key role in this regard. The strong attachment of EPS to the nanopatterns and the attempts of bacteria to move away from the unfavorable surface may lead to cell membrane damage. Moreover, mechanotransduction pathways in which the mechanical forces affect the metabolomics and the genomics of the bacteria could be possible mechanisms of bacterial death on the surface. © Elsevier. Reproduced by permission of Elsevier.
Consider, for instance, the existing problem of orthopedic implant-associated infections, a major cause of implant failure, and patients’ morbidity and even mortality. Different types of bacteria are able to attach to and grow on the implant surface, and eventually form a biofilm which impairs the function of the implant. One might say “We have been using antibiotics for a long time so what is the problem now? Kill all the bacteria before they form the biofilm”. It is not that simple. Leave aside the high costs of antibiotics and their efficient delivery routes which are sometimes associated with chemical alterations of the implant surface. The growing crisis of antibiotic resistance is a bigger obstacle. A previous solution has become a part of a new problem. Therefore, we need to look for alternative approaches.
Nature is a great source of inspiration in this regard. The wings of some insects such as cicada and dragonfly are covered with physical features with sizes at the nanoscale. The nanotopography of the wings can mechanically rupture the bacterial cell due to the sharp tips of the structures. Thanks to recent advances in nanofabrication techniques, it is now feasible to produce biomimetic surfaces with complex and precisely controlled nanotopography, also known as nanopatterns. Reactive ion etching (RIE), hydrothermal treatments, electron beam lithography (EBL), and nanoimprint lithography (NIL) being the most common fabrication methods, serve researchers in their quest for bactericidal nanopatterns. Using such fabrication methods, the surface of numerous materials can be patterned: silicon, titanium, and poly(methyl methacrylate) (PMMA) among others. But what characteristics should the nanopatterns possess to exhibit bactericidal properties?
Performing a systematic literature review, we tried to identify the effective range of dimensions that kill the bacteria and to discuss other determinant factors. We also intended to shed light on the underlying killing mechanisms of the nanopatterns. We realized that the dimensions of the majority of bactericidal nanopatterns investigated so far lie in the following range: 100 nm<height<1000 nm, 10 nm<diameter<300 nm, and spacing<500 nm. This range could be a bit narrower in case of specific types of nanopatterns such as nanopillars, nanocolumns, or nanocones. Uniformity and compaction of the nanopatterns, as well as the degree of bacterial attachment to the surface, are other factors influencing the killing efficiency. Direct penetration of nanopatterns into the cell wall and stretching of the bacterial cell between adjacent nanopatterns are among the proposed killing mechanisms of such surfaces (Fig. 1). Nanopatterns have another bright side too. They may promote host tissue regeneration by directing stem cells’ fate. Therefore, there seems to be a window within which bactericidal nanopatterns could also enhance bone tissue formation around the implant, guaranteeing its proper function.
So, is the next generation of orthopedic implants emerging? Hopefully yes. Nevertheless, researchers need more time to scrutinize it from different angles.
Khashayar Modaresifar
Department of Biomechanical Engineering, Faculty of Mechanical, Maritime, and Materials Engineering, Delft University of Technology, Mekelweg 2, 2628CD Delft, The Netherlands
Publication
Bactericidal Effects of Nanopatterns: A Systematic Review
Modaresifar K, Azizian S, Ganjian M, Fratila-Apachitei L, Zadpoor A
Acta Biomater. 2019 Jan 1













Leave a Reply
You must be logged in to post a comment.