Primary cilia: the story of the hair-like organelle that signals just became more complicated
Cells are the simplest functional unit of biological life. The basic components of cells are taught in biology classrooms across the globe, but despite being widely studied, cells still contain mysteries. Recently, scientists have rediscovered the existence of an organelle that was once thought to be a remnant of evolution, the primary cilium. Primary cilia are non-moving hair-like protrusions that extend from the top of cell surfaces where they are thought to act as cellular antennas as they participate in cellular communication. The surfaces of cilia are studded with receptors that can sense their environment. Importantly, primary cilia are now implicated in a variety of diseases, making it more necessary than ever to understand these structures.

Mutations in genes that result in changes in cilia function are thought to result in a variety of developmental disorders called ciliopathies. Recently, the role of primary cilia in disease was expanded to include chronic conditions with a recent discovery implicating cilia in prevention of atherosclerotic plaque formation related to heart disease and possible involvement in obesity. Given its importance in development, cell communication, and multiple human disorders, the study of primary cilia has attracted scientists from a wide range of fields. It is becoming more important than ever for scientists to clearly define the role of the primary cilium in disease, but this is easier said than done.
We now know of hundreds of different proteins that localize to the cilium (Fig. 1), to the base of the cilium (a structure called the basal body; Fig. 1), or to other structures that directly or indirectly result in defective cilia when certain genes are mutated. The story of this signaling organelle, however, has become more complex. These cilia proteins are increasingly being observed at cell areas that are not part of the cilium (Fig. 2). For instance, primary cilia proteins have been found in the cell nucleus where DNA is contained. They have been found at areas of the cell important for transport of proteins such as the Golgi body as well as at various intracellular vesicles and at cell borders. They can be found in mature cells and cells that are undergoing division (mitotic figures). They can even be found in specialized brain cell structures (i.e., growth cones and dendritic spines). Also, loss of “cilia genes” often results in more than just cilia problems, suggesting that these “cilia proteins” may have roles beyond the cilia (so called extraciliary functions). Interestingly, these “cilia proteins” can be found in cells that do not even have cilia (i.e., an immune system T-cell) suggesting many roles for these proteins.
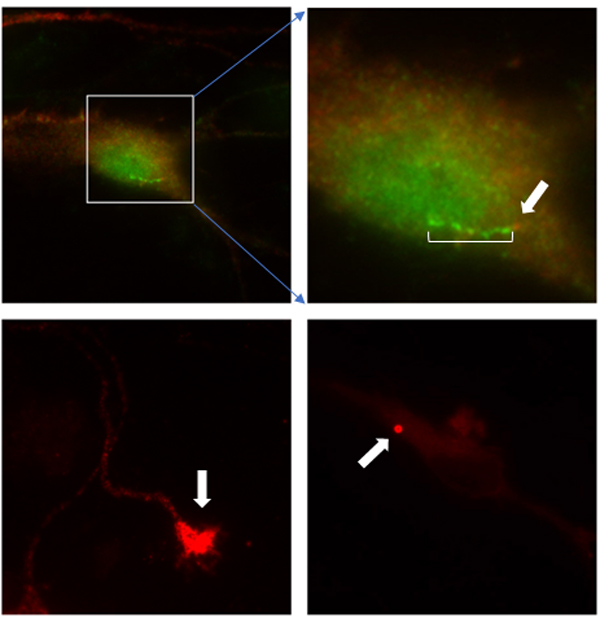
Such findings now complicate, or at the very least, introduce new considerations for cilia researchers when interpreting their results as to the true function of the cilium. If “cilia proteins” are found in cells that do not have cilia, is it correct to call them “cilia proteins” anymore? When a cilia gene is mutated, how are we sure that the resulting disease observed is due to abnormal cilia or is it due to abnormalities in one of the protein’s extraciliary functions or even both? It is an exciting time to be in the primary cilia field as we now have more complexities and questions than ever before that need to be answered.
Kiet Hua, Russell J. Ferland
Department of Neuroscience and Experimental Therapeutics, Albany Medical College, Albany, NY 12208, USA
Publication
Primary cilia proteins: ciliary and extraciliary sites and functions.Hua K, Ferland RJ
Cell Mol Life Sci. 2018 May













Leave a Reply
You must be logged in to post a comment.