Selecting for selective enzymes: Engineering P450s to produce chiral alcohols
Hydroxylation is the insertion of an oxygen atom into a C-H bond, to give chemicals with a C-O-H linkage (commonly known as alcohols). The process of hydroxylation is one difficult for chemists to achieve due to the low reactivity of the C-H bond, meaning that it is very stable and thus requires lot of energy to be activated. This energy input would typically derive from high temperature and /or high pressure chemical processes, often requiring the addition of expensive, toxic and even environmentally damaging metals and solvents. Despite this, hydroxylation reactions are known to occur in biology in the majority of living organisms. These organisms take a different strategy to chemists – using biological molecules, called enzymes, to lower the activation energy, thus allowing seemingly tricky reactions to occur at natural temperature, under atmospheric pressure and in the absence of man-made chemicals and catalysts.

The enzymes responsible for hydroxylation in biological systems are called cytochrome P450s. These enzymes work by using oxygen as it is found in the air and selecting a particular C-H bond on a specific molecule for insertion. The ability of P450s to insert at a single position into only one of the many CH bond on a molecule makes them selective. In this work researchers have sought to use a P450s to form a single hydroxyl group selectively on methylethylbenzene (shown below). Using a cytochrome P450 called RhF from the bacterial genus Rhodococcus, it was found that the hydroxylation reaction was catalysed selectively at either of two particular C-H bonds in one region of the molecule (regioselectivity). Despite this excellent catalyst control, the occurrence of the reaction at the C-H bonds on either side of the molecule at the specific position created a mixture of mirror image products (enantiomers).
In an attempt to address this low enantioselectivity (distinguishing between two otherwise identical chemical groups on opposite sides of a molecule at a single position) several steps were taken. Using the structure of the enzyme and the shape of the 2-methyl-ethylbenzene starting material, computer modelling was used to predict the way these two interact. Using in-depth knowledge of how the P450 hydroxylation is known to proceed, the model could be used to predict how changes in the shape of the enzyme could alter the way it interacted with the compound of interest and thus which C-H bond was more likely to be activated. Using these predictions as a guide, the enzyme was modified using genetic engineering techniques in accordance with the computational model. The gene encoding the enzyme was isolated and mutated to change its code and thus alter the structure and properties of the enzyme produced. Several different mutations were made at various positions in the gene to create a library of DNA constructs which were then used to genetically modify laboratory strain bacteria. By subjecting these to certain conditions, distinct colonies of bacteria could be grown such that each colony contained a single mutant gene and thus produced only a single variant of the enzyme. To check that the mutated genes gave enzymes which still functioned correctly, a visual screen was conducted using a compound called indole which is known to form an indigo-coloured dye when acted upon by P450 enzymes. This was done using colonies of bacteria such that those colonies which changed colour and thus harboured an active mutant gene, could be selected for further investigation.
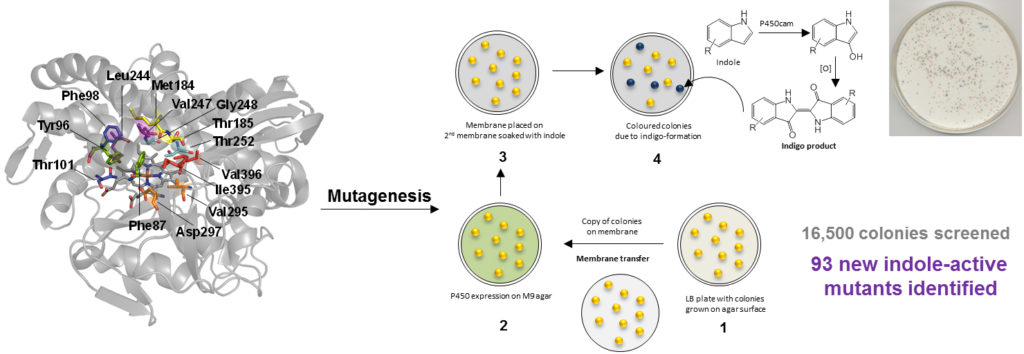 The DNA from the colonies shown to have the P450’s colour changing activity was extracted and introduced into new, separate batches of bacteria. These were then grown on a large scale and used as whole cells containing each newly designed enzyme to catalyse methyl-ethylbenzene hydroxylation. After the reaction, the distribution of the two mirror image products was measured and it was found that several of the redesigned enzymes gave much better enantioselectivity, with some reacting exclusively with one of the two opposing C-H bonds. This work provides insights into the mechanisms by P450 enzymes selectively activate C-H bonds and has allowed creation of new enzyme variants which may one day be used to perform industrially-relevant reactions without high energy input and associated environmental damage.
The DNA from the colonies shown to have the P450’s colour changing activity was extracted and introduced into new, separate batches of bacteria. These were then grown on a large scale and used as whole cells containing each newly designed enzyme to catalyse methyl-ethylbenzene hydroxylation. After the reaction, the distribution of the two mirror image products was measured and it was found that several of the redesigned enzymes gave much better enantioselectivity, with some reacting exclusively with one of the two opposing C-H bonds. This work provides insights into the mechanisms by P450 enzymes selectively activate C-H bonds and has allowed creation of new enzyme variants which may one day be used to perform industrially-relevant reactions without high energy input and associated environmental damage.
Nicholas J. Weise
Manchester Institute of Biotechnology,
University of Manchester, Manchester, UK
Publication
Enantioselective Benzylic Hydroxylation Catalysed by P450 Monooxygenases: Characterisation of a P450cam Mutant Library and Molecular Modelling.
Eichler A, Gricman Ł, Herter S, Kelly PP, Turner NJ, Pleiss J, Flitsch SL
Chembiochem. 2016 Mar 2













Leave a Reply
You must be logged in to post a comment.