Epstein–Barr virus in peripheral blood predicts response to rituximab therapy in rheumatoid arthritis
Rheumatoid arthritis (RA) is an autoimmune disease, characterized by chronic inflammation of the joints that may cause permanent cartilage and bone damage. The cause of RA is unknown. It is, however, believed that a combination of genes and environmental factors – such as infection with certain viruses and bacteria – may trigger the disease.
The Epstein-Barr virus (EBV), is one of eight viruses in the herpes family, and is one of the most common viruses in humans. It is best known as the cause of infectious mononucleosis (glandular fever). After infection of the oropharynx and nasopharyngeal tissues, EBV infects B-cells, a type of white blood cell in the immune system. Within the B-cells the virus persists in a partially replicated state for the life of the cell. Genetic susceptibility to the effects of B-cell infection by EBV leads to an increased number of autoreactive B cells which are hypothesized to cause autoimmune diseases including RA. Autoreactive B-cells produce antibodies that target our own tissues
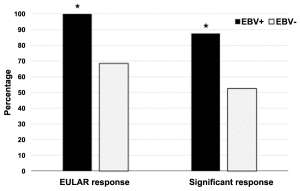
Fig. 1. Proportion of RA patients achieving good/moderate EULAR response and significant response (DAS28 change>1.2) after RTX. *P less than 0.05 for EBV positive vs. EBV negative patients
Rituximab (RTX), (also known as Mabthera or Rituxan) is a protein which binds to the surface of B-cells resulting in their destruction and is therefore used to treat diseases which are characterized by excessive numbers of B cells, overactive B cells, or dysfunctional B cells. This includes many lymphomas, leukemias, and autoimmune disorders. RTX is used to treat RA that has not improved with other types of medications.
The aim of our study was to evaluate whether EBV genome detected in white blood cells collected prior to rituximab treatment would predict response to B-cell depleting therapy with RTX in RA.
35 RA patients who started treatment with rituximab (RTX) were assessed for baseline disease activity using 28-joint Disease Activity Score (DAS28). DAS28 combines scores on a 28-tender and swollen joint count, a patient-reported measure for general health, and an inflammatory marker (either the erythrocyte sedimentation rate or the C-reactive protein) into a composite measure of disease activity in RA. Treatment response was evaluated 3 – 7 months after RTX.
EBV DNA was detected in 16/35 (46 %) of the baseline blood samples. At baseline the EBV DNA positive and EBV negative patients were not different in terms of age, disease activity or disease duration. All 16 EBV DNA positive patients achieved at least a moderate response to RTX as determined by decline in DAS28 score. Although more than 50 % of the EBV negative RA patients also responded to RTX, the proportion of responders was statistically significantly larger in the EBV positive patient group (Fig. 1).
Our results suggest that EBV genome in peripheral blood could be used as a predictive marker for response to RTX in the treatment of RA. Our findings also support the hypothesis that EBV might contribute to onset or perpetuation of the autoimmune response in RA and suggest future rationale for preventive and therapeutic measures for RA, such as EBV vaccination or regulation of the existing EBV infection by the use of antiviral agents.
Publication
Epstein-Barr virus in peripheral blood is associated with response to rituximab therapy in rheumatoid arthritis patients.
Valleala H1, Kauppi MJ, Kouri VP, Korpela M.
Clin Rheumatol. 2015 Aug
Related Articles:
 | Unlocking new treatments for bone diseases: using… Despite appearances, bones are a constantly changing organ in the body. Cells that produce new bone (named osteoblasts) and cells that break down bone (named osteoclasts) work in harmony to… |
 | PEPITEM supports new bone and blood vessel growth in… While bones may appear solid and unchanging, they are a living tissue that is constantly being broken down and rebuilt in a process known as bone remodelling. Specialised cells (called… |
 | Is multiple sclerosis triggered by immunological… Multiple sclerosis (MS) is an autoimmune disease where immune cells (T cells) and antibodies progressively damage the myelin sheath surrounding nerve cells leading to their loss of function. We have… |
 | Improving assessment of arthritis models to better… Rheumatoid Arthritis (RA) is a common inflammatory disease that is characterized by swelling and tenderness of multiple joints. The resulting pain and joint stiffness cause disability for patients and treatment… |
 | Looking inside the heart: how multiple chronic… The aim of this study was to understand how having several ongoing health problems—what we refer to as multimorbidity—impacts the heart in people with cardiovascular disease, especially those undergoing heart… |
 | Naturally occurring peptide may tackle the “root… Obesity typically occurs when a person eats more calories than they need for their daily activity. It results in a build-up of fatty tissue around all organs. In lean people,… |







Leave a Reply
You must be logged in to post a comment.