Brain-derived neurotrophic factor is a miracle fertilizer for our brain?
Brain-derived neurotrophic factor (BDNF) is sometimes called “miracle grow for the brain” because several studies have reported that BDNF “nourishes neurons like fertilizer.” It is well-known that BDNF protects brain cells and stimulates growth of their dendrites and axons. In addition, BDNF has been associated with enhancement of learning and cognitive function.
However, BDNF does not always play a positive role for our brain health.

Fig. 1. School bullying (top) and defeat stress in mouse model (bottom, Courtesy, with permission: Georgia Hodes. Photos by Christopher Wood)
Social stress is one of the most critical factors in the onset of depressive disorders in humans. A long-lasting aversion to social contact produced by repeated aggression in rodents has been proposed as a model of chronic stress-induced depression in humans. The effect of social stress on depressive-like behavioral abnormalities has been investigated with the chronic social defeat stress paradigm in mice, in which susceptible and resilient phenotypes are segregated after 10 days of the defeat stress. Depressive-like behaviors in susceptible mice have been causally associated with molecular and physiological abnormalities in the mesolimbic dopamine (DA) pathway, which is comprised of the ventral tegmental area (VTA) dopamine neurons and their projecting terminals to the nucleus accumbens (NAc).
Contrary to popular belief, BDNF in the mesolimbic DA pathway plays an aversive role in the susceptible phenotype after chronic social defeat stress. Elevated levels of BDNF protein expression in NAc are associated with depressive-like abnormalities induced by chronic social defeat stress, and are not observed in resilient mice. Localized Bdnf gene deletion in VTA of adult mice reduces susceptibility to chronic social defeat stress, suggesting that BDNF, transported from VTA to NAc, induces behavioral susceptibility.
Of note, light-induced optogenetic activation of the VTA-to-NAc mesolimbic DA pathway, which mimics enhanced burst firings in the susceptible mice, facilitates release of BDNF as well as DA from VTA DA terminals. BDNF can also facilitate DA release from DA terminals. A subset of VTA DA neurons has been also implicated in stress-elicited depressive-like abnormalities. Thus, it is conceivable that both DA and BDNF signaling in NAc might promote depressive phenotypes. However, it is still not clear if DA, BDNF, or both are important for stress-elicited depressive-like phenotypes.
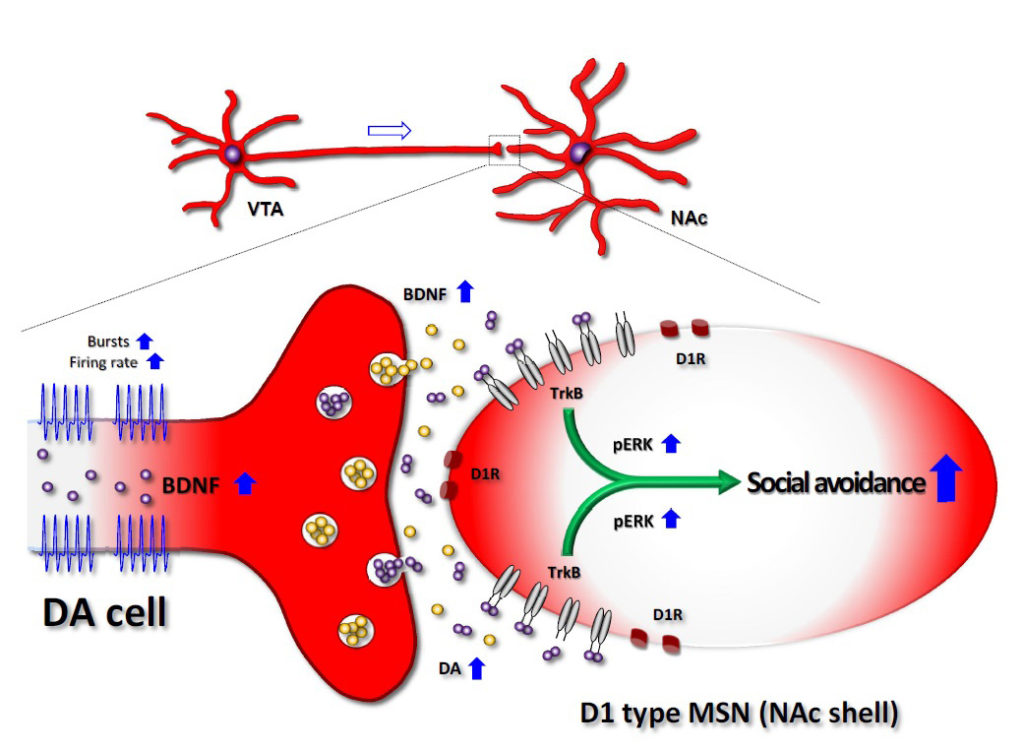
Fig 2. Model for mesolimbic BDNF action in the chronic defeat stress-induced social avoidance behavior. Inset, reward circuitry in human brain.
Koo et al. demonstrate that BDNF-TrkB signaling, but not DA signaling, in the mesolimbic DA pathway is critical for the susceptible phenotype that is induced by chronic defeat stress. BDNF-TrkB blockade into NAc reversed chronic defeat-induced social avoidance behavior but DA antagonists did not. Optogenetic activation of VTA-to-NAc mesolimbic DA pathway exacerbated the chronic defeat-induced social avoidance behavior. The aggravated social avoidance behavior by the repeated optogenetic activation in chronic defeats was totally blocked by BDNF-TrkB blockade into NAc and by localized BDNF depletion in VTA. Interestingly, DA release was not changed by the chronic defeat events nor by additional optogenetic activation during chronic social defeat.
These findings validate a crucial role of the mesolimbic BDNF-TrkB signaling in facilitating depressive-like outcomes and a potential therapeutic path for depression treatment. However, this pro-depressant role of mesolimbic BDNF signaling is in direct contrast to the antidepressant-like actions of BDNF in hippocampus, which emphasizes the circuit-specific nature of molecular mechanisms involved in brain disease and the need to identify therapeutic approaches that take such circuit specificity into consideration.
Ja Wook Koo
Fishberg Department of Neuroscience, Friedman Brain Institute, Icahn School of Medicine at Mount Sinai,
New York, New York, USA
Department of Neural Development and Disease, Korea Brain Research Institute,
Daegu, Republic of Korea
Publication
Essential Role of Mesolimbic Brain-Derived Neurotrophic Factor in Chronic Social Stress-Induced Depressive Behaviors.
Wook Koo J, Labonté B, Engmann O, Calipari ES, Juarez B, Lorsch Z, Walsh JJ, Friedman AK, Yorgason JT, Han MH, Nestler EJ
Biol Psychiatry. 2015 Dec 15
Related Articles:
 | Social rank recognition is altered in autism… Humans and many other animals spend their lives in social groups, along with constructing social hierarchy within the groups. Since social hierarchy is a principal determinant for allocation of limited… |
 | Making Christmas trees under duress, or how cells… Some of the most enduring images for a molecular biologist are electron microscopy micrographs of the so-called “Christmas trees”, famously first observed by Oscar Miller from newt oocytes in 1969.… |
 | Rabbits with mammary carcinomas as a model for… Within a breeding colony of rabbits, the American pathologist Harry Greene (1904-1969) observed that mammary carcinomas were restricted to certain families. This is suggestive of a familiar predisposition as it… |
 | Boosting chicken health with tea extract: a natural… In modern poultry farming, chickens often face stressful conditions such as overcrowding, poor air quality, and contaminated feed. These conditions can lead to oxidative stress, a state where an excess… |
 | Looking inside the heart: how multiple chronic… The aim of this study was to understand how having several ongoing health problems—what we refer to as multimorbidity—impacts the heart in people with cardiovascular disease, especially those undergoing heart… |
 | Fatigue assessment strategy using Bayesian techniques The fulfilment of the probabilistic fatigue characterization of materials based on the S-N field requires the definition of the statistical distribution of the percentile curves of the failure probability, interpreted… |







Leave a Reply
You must be logged in to post a comment.