Helicase and ribosome share a common mechanism of nucleic acid unwinding
In the cell DNA is present in the duplex form and many messenger RNAs (mRNAs) can form duplex structures. To access the genetic information (represented by nucleotides or codons) on the single-stranded (ss) nucleic acids (NA) by enzymes for replication, repair or translation, the duplex must be separated. Helicase is responsible for unwinding of NA duplex and ribosome that is responsible for translation of mRNA into protein can also unwind mRNA duplex. Here, it is proposed that helicase and ribosome use a common mechanism to unwind NA duplex. In the model, the main players are the enzyme’s two domains or subunits called A and B (Fig. 1).
We begin the chemomechanical coupling cycle with A binding strongly to ssNA while B having an intermediate interaction with ssNA (Fig. 1a). The change in the nucleotide state induces a conformational change of the enzyme, changing the distance between A and B. Since now A has strong interaction with ssNA, the change in the distance induces the movement of B relative to the ssNA (Fig. 1b). The conformational change or the further change in the nucleotide state induces the change in the affinity of A (Fig. 1c). The subsequent reverse conformational change of the enzyme changes reversely the distance between A and B. Since now A has weak interaction with ssNA, the reverse change in the distance induces the downstream movement of A with B being fixed to the ssNA (Fig. 1d). In the presence of downstream duplex, the movement of A would induce NA unwinding. However, the resistance resulting from the NA unwinding would impede the downstream movement of A and thus the reverse change in the distance between A and B could also induce the upstream movement of B by overcoming the intermediate binding energy of B with the ssNA (Fig. 1e). The reverse conformational change then induces the reverse change in the affinity of A (Fig. 1f or a).
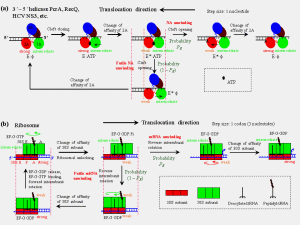
Fig. 2. For helicase, Fig. 1 is concretely represented by transitions in (a). For ribosome, Fig. 1 is concretely represented by transitions in (b).
For the helicase that has a structure consisting of four domains, Fig. 1 is concretely represented in Fig. 2a, where domains 2A and 1A correspond to A and B, respectively. ATP binding induces closing of the left between domains 2A and 1A, in turn changing the binding affinity of domain 2A to the ssNA from strong to weak. The release of ATP-hydrolysis products induces opening of the cleft, in turn changing the binding affinity of domain 2A to the ssNA from weak to strong. For the bacterial ribosome, Fig. 1 is concretely represented in Fig. 2b, where subunits 30S and 50S correspond to A and B, respectively. Subunit 50S interacts with mRNA via two tRNAs that form codon-anticodon interactions with the mRNA and subunit 30S binds directly to the mRNA. The binding of elongation factor G complexed with GTP () and then GTP hydrolysis in the rotated ribosome weakens the interaction between subunit 30S and mRNA (called ribosomal unlocking). The subsequent reverse intersubunit rotation causes subunit 30S to move relative to the mRNA by one codon. In the non-rotated ribosome the interaction of subunit 30S with mRNA becomes strong.
With the model, a lot of experimental data on NA unwinding by the helicase and ribosome can be explained well.
Ping Xie
Laboratory of Soft Matter Physics, Institute of Physics, CAS, China
Publication
A unified model of nucleic acid unwinding by the ribosome and the hexameric and monomeric DNA helicases.
Xie P
J Theor Biol. 2015 Sep 7














Leave a Reply
You must be logged in to post a comment.