Urine lipoarabinomannan assays for paediatric tuberculosis
Tuberculosis is one of the most common infectious diseases worldwide with 2 billion people being latently infected and 9 million people diagnosed with active disease every year. Infection with TB is also a major contributor to childhood morbidity and mortality, estimating that children account for as much as 10-20% of the total TB burden in a TB endemic country such as Tanzania. The diagnosis of tuberculosis (TB) is usually established by detecting mycobacteria, either through smear microscopy or culturing methods. However, the diagnosis based on the detection of the bacilli is extremely difficult, especially in children who cannot produce sputum so easily. Lipoarabinomannan (LAM) detection in urine for TB diagnosis was first investigated in the late 1990s. LAM is a 19 kilodalton (± 8.5 kD) lipopolysaccharide, specific to the cell wall of the genus Mycobacterium and is released from metabolically active or degrading bacterial cells. It can subsequently be detected in urine and other body fluids.
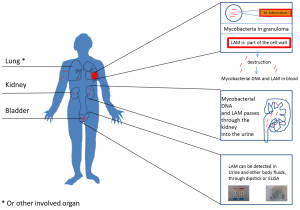 Advantages of urine LAM diagnosis include the ease of specimen collection, short and simple testing algorithm, low cost and relatively low training and set up requirements. Ideally the LAM-strip assay can be performed as a point-of-care test in remote settings.
Advantages of urine LAM diagnosis include the ease of specimen collection, short and simple testing algorithm, low cost and relatively low training and set up requirements. Ideally the LAM-strip assay can be performed as a point-of-care test in remote settings.
We evaluated the diagnostic performance of two commercially available assays, the MTB-LAM-ELISA (Chemogen, Portland USA) (an assay requiring a laboratory) and the Determine TB-LAM (Alere, Waltham, MA, US) (a bedside test) in Tanzanian children with presumed TB and a high HIV co-infection rate.
In a prospective study, 132 children with suspected active TB were assigned to classified diagnostic subgroups. Clinical examination, sputum microscopy and culture, Xpert MTB/RIF assay (a fairly new, fully automated technique for the diagnosis of TB), as well as HIV testing was performed. Urine samples were subjected to testing by both LAM assays to ascertain their sensitivity and specificity.
The overall HIV prevalence in our study cohort was 51%. Using culture as the diagnostic gold standard, eighteen children had culture-confirmed pulmonary tuberculosis, 10 of them were co-infected with HIV. The number of culture-confirmed cases seems low, only 13.6%, but is typical for the diagnostic difficulties of childhood TB.
 Focusing on the proven TB cases, the sensitivity for both Lam diagnostic tests was higher in HIV-positive compared to HIV-negative children. In HIV positive children a sensitivity of 70% was found for the MTB-LAM-ELISA and 50% for Determine TB-LAM. Smear microscopy, still the most commonly used technique worldwide, could only find 30% of the culture-confirmed HIV/TB co-infected cases. In HIV negative children both LAM tests performed very poorly, with a sensitivity of 13% for the MTB-LAM-and 0% for Determine TB-LAM. The superior performance in HIV-positive individuals is in contrast to other methods where performance is worse in HIV/TB co-infected patients.
Focusing on the proven TB cases, the sensitivity for both Lam diagnostic tests was higher in HIV-positive compared to HIV-negative children. In HIV positive children a sensitivity of 70% was found for the MTB-LAM-ELISA and 50% for Determine TB-LAM. Smear microscopy, still the most commonly used technique worldwide, could only find 30% of the culture-confirmed HIV/TB co-infected cases. In HIV negative children both LAM tests performed very poorly, with a sensitivity of 13% for the MTB-LAM-and 0% for Determine TB-LAM. The superior performance in HIV-positive individuals is in contrast to other methods where performance is worse in HIV/TB co-infected patients.
Regarding specificity, no LAM positive urine was found among children where TB could be reliable excluded. In a previous study false positive LAM results had been observed, most likely due to contamination of the sample with environmental (myco-) bacteria. In this study urine samples were collected with great precautions, including washing instructions and the use of clean containers, in order to avoid false positive result
In conclusion, both LAM tests demonstrated a reasonable sensitivity in HIV-positive TB infected children, whereas the sensitivity in HIV-negative children was extremely poor. This holds promise for accelerated TB-diagnosis in HIV co-infected children which might in turn have a positive impact on TB related childhood morbidity and mortality. The combination of LAM tests with other rapid TB diagnostics, such as smear microscopy, could substantially improve the overall detection of TB in HIV infected and uninfected children, as different methods perform well under different circumstances.
Publication
Performance of urine lipoarabinomannan assays for paediatric tuberculosis in Tanzania.
Kroidl I, Clowes P, Reither K, Mtafya B, Rojas-Ponce G, Ntinginya EN, Kalomo M, Minja LT, Kowuor D, Saathoff E, Kroidl A, Heinrich N, Maboko L, Bates M, O’Grady J, Zumla A, Hoelscher M, Rachow A.
Eur Respir J. 2015 Sep













Leave a Reply
You must be logged in to post a comment.