Ultrasensitive enzyme immunoassay by immune complex transfer method
Many technologies have been developed to improve the sensitivity of immunoassays, such as high affinity antibodies, specific conjugations using the hinge region of Fab’ fragments, highly active enzymes, and digital counting systems. We described a methodology called Immune Complex Transfer-Enzyme Immunoassay (ICT-EIA) in the article published in Journal of immunological methods in 2018.
ICT-EIA uses two types of beads and sandwich immune complexes are transferred from the 1st bead to the 2nd bead in the assay (Fig. 1). In a typical immunoassay, the detection antibody may be nonspecifically bound to the bead surface emitting a background signal, which may prevent higher sensitivity. ICT-EIA is a technology that improves the sensitivity of immunoassays by reducing this non-specific binding.
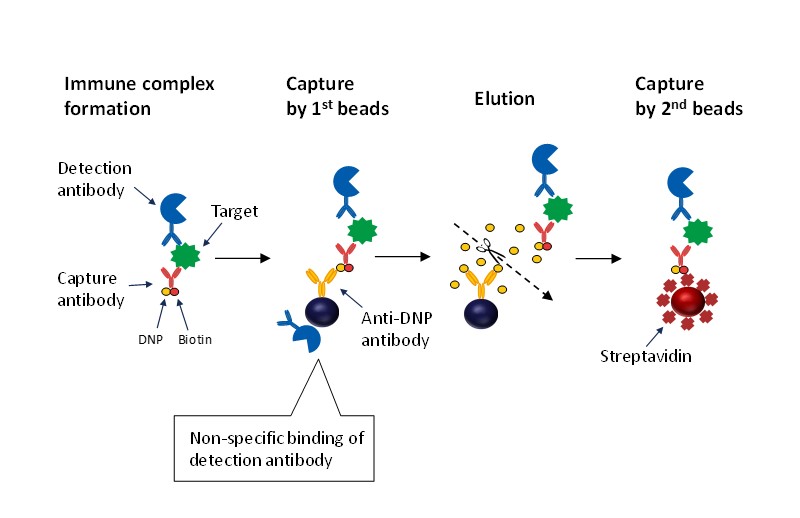
Fig. 1. Assay flow of ICT-EIA. ICT-EIA can reduce the amount of detection antibodies non-specifically bound to the bead surface by transferring immune complexes to another bead.
The article was written mainly for two purposes, 1) to clarify the sensitivity level of ICT-EIA by direct comparison with sandwich EIA (Sand-EIA) and 2) to reveal the mechanism of the high sensitivity by step-by-step analysis of the reaction steps of ICT-EIA.
We developed both Sand-EIA and ICT-EIA for three cytokines, TNF-α, TARC, and IL-12/23p40. All antibodies and antigens were purchased from R&D systems (Minneapolis, MN). Fab’ fragments of these antibodies were conjugated with β-D-galactosidase as detection reagent and with dinitrophenyl-biotinyl-BSA (DNP-Bio-BSA) as capture reagent, respectively. Whole IgG labeled with DNP and Biotin was also used as capture antibody in TNF-α assay. The same conjugates were used for both Sand-EIAs and ICT-EIAs.
1) Improved sensitivities of ICT-EIAs for cytokines
Sand-EIAs and ICT-EIAs were performed for cytokines (Fig. 2). The limit of detection (LoD) was defined as the minimum amount of antigen that gives a significant bound enzyme activity (fluorescence signal) compared with the background signal (no antigen). A significant difference from the background was confirmed using a t-test (P < 0.05). The LODs of Sand-EIA/ICT-EIA for TNF-α, TARC, and IL-12/23p40 were 3/0.03, 1/0.01, and 3/0.03 pg/mL, respectively. ICT-EIAs had 100 times higher sensitivity than Sand-EIAs when the same buffers, antibody conjugates, and polystyrene beads were used.
2) The mechanism of improved sensitivity by ICT-EIA

Fig. 2. Dose response curves for cytokines. ICT-EIAs had 100 times higher sensitivity than Sand-EIAs.
To reveal the mechanism of signal transfer in ICT-EIA, the enzyme activity of the detection conjugate was measured after capture on the 1st bead, after elution from the 1st bead, and after capture on the 2nd bead using a fluorometric assay. In the case of the TNF-α assay, the enzymatic activity generated by the sandwich-immune complex captured on the 1st bead was mostly released (87%) into the eluate by the DNP-Lys used in the elution step, but not all (34%) could be re-captured by the 2nd bead. This reduction is an adverse effect of the immune complex transfer method. Elution of the sandwich-immune complex from the 1st bead proceeded ideally, but undesirable unbinding of the Ag-Ab sandwich-immune complex occurred in the eluate because there was no excess of antibody to drive the reaction after the elution. On the other hand, the enzyme activity from non-specifically bound detection antibody conjugate improved by 280-fold compared to the 1st bead. The biggest difference in binding conditions between the 1st bead and the 2nd bead is the ratio of detection antibody conjugate to immune complex. The ratio was about 1.5 × 104 during the 1st capture reaction (at 3 pg/mL of TNF-α EIA (LoD of Sand-EIA)) but only 1.5 during the 2nd capture reaction. ICT-EIA was more sensitive than Sand-EIA due to greatly reduced non-specific signal levels, despite exhibiting somewhat reduced specific signal levels.
Toshihiro Watanabe
Clinical Innovation, Sysmex Corporation, 4-4-4 Takatsukadai, Nishi-ku, Kobe, Japan
Publication
The immune complex transfer enzyme immunoassay: Mechanism of improved sensitivity compared with conventional sandwich enzyme immunoassay.
Watanabe T, Hashida S
J Immunol Methods. 2018 Aug













Leave a Reply
You must be logged in to post a comment.